HYDROXYZINE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Hydroxyzine, and what generic alternatives are available?
Hydroxyzine is a drug marketed by Baxter Hlthcare, Altana, Am Regent, Dr Reddys, Fresenius Kabi Usa, Hospira, Pharmafair, Smith And Nephew, Solopak, Watson Labs, Wyeth Ayerst, Alpharma Us Pharms, Anima, Apozeal Pharms, Chartwell Rx, Hikma, Kv Pharm, Lannett Co Inc, Pai Holdings Pharm, Able, Actavis Elizabeth, Amneal Pharm, Aurobindo Pharma Ltd, Epic Pharma Llc, Graviti Pharms, Halsey, Heritage Pharma, Hetero Labs Ltd Iii, Invagen Pharms, Ivax Pharms, Kvk Tech, Mutual Pharm, Northstar Hlthcare, Nuvo Pharms Inc, Pliva, Prinston Inc, Purepac Pharm, Quantum Pharmics, Rising, Sandoz, Strides Pharma, Sun Pharm Inds Inc, Sun Pharm Industries, Superpharm, Usl Pharma, Barr, Beximco Pharms Usa, Duramed Pharms Barr, Impax Labs Inc, Ivax Sub Teva Pharms, Par Pharm, and Vangard. and is included in one hundred and thirty-four NDAs.
The generic ingredient in HYDROXYZINE is hydroxyzine pamoate. There are nineteen drug master file entries for this compound. Twenty-nine suppliers are listed for this compound. Additional details are available on the hydroxyzine pamoate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Hydroxyzine
A generic version of HYDROXYZINE was approved as hydroxyzine pamoate by SANDOZ on December 31st, 1969.
Summary for HYDROXYZINE
| US Patents: | 0 |
| Applicants: | 52 |
| NDAs: | 134 |
| Raw Ingredient (Bulk) Api Vendors: | 71 |
| Clinical Trials: | 32 |
| Patent Applications: | 4,164 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for HYDROXYZINE |
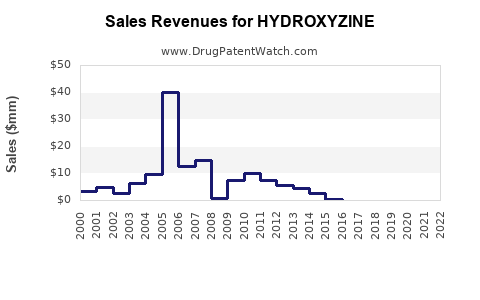
| Drug Sales Revenues: | Drug sales revenues for HYDROXYZINE |
| DailyMed Link: | HYDROXYZINE at DailyMed |
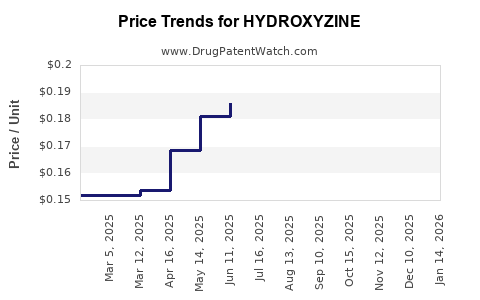
See drug prices for HYDROXYZINE
Recent Clinical Trials for HYDROXYZINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Sultan Qaboos University | Phase 4 |
| Minia University | Phase 1 |
| Mongi Slim Hospital | N/A |
Anatomical Therapeutic Chemical (ATC) Classes for HYDROXYZINE
US Patents and Regulatory Information for HYDROXYZINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ivax Pharms | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | TABLET;ORAL | 087216-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Quantum Pharmics | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | TABLET;ORAL | 088540-001 | Oct 22, 1985 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Able | HYDROXYZINE HYDROCHLORIDE | hydroxyzine hydrochloride | TABLET;ORAL | 040563-001 | Jul 22, 2004 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


