Amarin Pharms Company Profile
✉ Email this page to a colleague
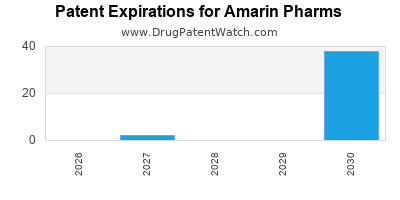
What is the competitive landscape for AMARIN PHARMS, and when can generic versions of AMARIN PHARMS drugs launch?
AMARIN PHARMS has two approved drugs.
There are seventy US patents protecting AMARIN PHARMS drugs.
There are three hundred and eight patent family members on AMARIN PHARMS drugs in forty-five countries and nineteen supplementary protection certificates in twelve countries.
Drugs and US Patents for Amarin Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | 8,415,335 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-001 | Jul 26, 2012 | AB | RX | Yes | Yes | 8,617,594 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | 10,842,768 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | AB | RX | Yes | No | 10,278,935 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Amarin Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-001 | Jul 26, 2012 | 8,188,146 | ⤷ Try a Trial |
| Amarin Pharms | VASCEPA | icosapent ethyl | CAPSULE;ORAL | 202057-002 | Feb 16, 2017 | 8,188,146 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for AMARIN PHARMS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Capsules | 1 g | ➤ Subscribe | 2013-01-15 |
| ➤ Subscribe | Capsules | 500 mg | ➤ Subscribe | 2017-08-29 |
| ➤ Subscribe | Capsules | 1 g | ➤ Subscribe | 2016-07-26 |
International Patents for Amarin Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Chile | 2014003534 | ⤷ Try a Trial |
| Slovenia | 3363433 | ⤷ Try a Trial |
| Colombia | 6470838 | ⤷ Try a Trial |
| European Patent Office | 2866801 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Amarin Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2443246 | 39/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: ICOSAPENT-ETHYL (= EICOSAPENTAENSAEUREETHYLESTER,; REGISTRATION NO/DATE: EU/1/20/1524 20210329 |
| 2443246 | PA2021522 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: IKOZAPENTO ETILAS; REGISTRATION NO/DATE: EU/1/20/1524 20210326 |
| 2022495 | 132021000000154 | Italy | ⤷ Try a Trial | PRODUCT NAME: ICOSAPENT ETILE(VAZKEPA); AUTHORISATION NUMBER(S) AND DATE(S): EU/1/20/1524, 20210329 |
| 0113964 | 97C0037 | Belgium | ⤷ Try a Trial | PRODUCT NAME: OESTROGENES EQUINS CONJUGUES; ACETATE DE MEDROXYPROGESTERONE; NAT. REGISTRATION NO/DATE: NL 19569 19950301; FIRST REGISTRATION: CH - 52647 01 010 19940826 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.