Astellas Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ASTELLAS, and when can generic versions of ASTELLAS drugs launch?
ASTELLAS has thirty-seven approved drugs.
There are twenty-five US patents protecting ASTELLAS drugs. There is one tentative approval on ASTELLAS drugs.
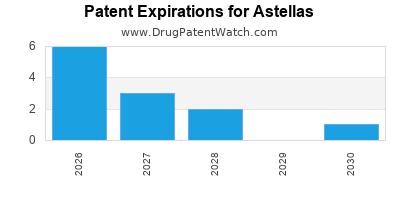
There are four hundred and thirty-three patent family members on ASTELLAS drugs in forty-two countries and sixty-five supplementary protection certificates in nineteen countries.
Summary for Astellas
| International Patents: | 433 |
| US Patents: | 25 |
| Tradenames: | 21 |
| Ingredients: | 16 |
| NDAs: | 37 |
| Patent Litigation for Astellas: | See patent lawsuits for Astellas |
Drugs and US Patents for Astellas
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Astellas | CRESEMBA | isavuconazonium sulfate | POWDER;INTRAVENOUS | 207501-001 | Mar 6, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Astellas | CEFIZOX | ceftizoxime sodium | INJECTABLE;INJECTION | 050560-001 | Sep 15, 1983 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Astellas | IZERVAY | avacincaptad pegol sodium | SOLUTION;INTRAVITREAL | 217225-001 | Aug 4, 2023 | RX | Yes | Yes | 8,236,773 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Astellas | VESICARE LS | solifenacin succinate | SUSPENSION;ORAL | 209529-001 | May 26, 2020 | RX | Yes | Yes | 9,918,970 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astellas | XTANDI | enzalutamide | TABLET;ORAL | 213674-001 | Aug 4, 2020 | RX | Yes | No | 11,839,689 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Astellas | PROGRAF | tacrolimus | FOR SUSPENSION;ORAL | 210115-001 | May 24, 2018 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Astellas
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Astellas | ASTAGRAF XL | tacrolimus | CAPSULE, EXTENDED RELEASE;ORAL | 204096-001 | Jul 19, 2013 | 6,884,433 | ⤷ Try a Trial |
| Astellas | ASTAGRAF XL | tacrolimus | CAPSULE, EXTENDED RELEASE;ORAL | 204096-001 | Jul 19, 2013 | 8,551,522 | ⤷ Try a Trial |
| Astellas | LEXISCAN | regadenoson | SOLUTION;INTRAVENOUS | 022161-001 | Apr 10, 2008 | 9,045,519 | ⤷ Try a Trial |
| Astellas | ADENOSCAN | adenosine | SOLUTION;INTRAVENOUS | 020059-001 | May 18, 1995 | 5,070,877 | ⤷ Try a Trial |
| Astellas | ASTAGRAF XL | tacrolimus | CAPSULE, EXTENDED RELEASE;ORAL | 204096-002 | Jul 19, 2013 | 6,440,458 | ⤷ Try a Trial |
| Astellas | MYCAMINE | micafungin sodium | INJECTABLE;INTRAVENOUS | 021506-002 | Mar 16, 2005 | 6,107,458 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ASTELLAS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Capsules | 0.5 mg, 1 mg, and 5 mg | ➤ Subscribe | 2013-11-15 |
| ➤ Subscribe | For Injection | 50 mg/vial and 100 mg/vial | ➤ Subscribe | 2014-06-16 |
| ➤ Subscribe | Injection | 3 mg/mL, 20 mL and 30 mL vials | ➤ Subscribe | 2005-04-16 |
| ➤ Subscribe | Tablets | 5 mg and 10 mg | ➤ Subscribe | 2009-04-08 |
| ➤ Subscribe | Capsules | 40 mg | ➤ Subscribe | 2016-08-31 |
| ➤ Subscribe | Injection | 3 mg/mL, 20 mL and 30 mL vials | ➤ Subscribe | 2005-04-18 |
| ➤ Subscribe | Injection | 0.08 mg/mL, 5 mL vial | ➤ Subscribe | 2012-04-10 |
International Patents for Astellas Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 2015003140 | ⤷ Try a Trial |
| Luxembourg | 92939 | ⤷ Try a Trial |
| Brazil | PI0015254 | ⤷ Try a Trial |
| Denmark | 3318259 | ⤷ Try a Trial |
| China | 110003114 | ⤷ Try a Trial |
| Japan | 2013010791 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Astellas Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1189916 | 122011000010 | Germany | ⤷ Try a Trial | PRODUCT NAME: REGADENOSON UND DIE SALZE DAVON; REGISTRATION NO/DATE: EU/1/10/643/001 20100906 |
| 2428508 | SPC/GB20/007 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: GILTERITINIB OR A SALT THEREOF; REGISTERED: UK EU/1/19/1399(FOR NI) 20191028; UK PLGB00166/0425 20191028 |
| 1893196 | CR 2013 00065 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ENZALUTAMID ELLER ET FARMACEUTISK ACCEPTABELT SALT DERAF; REG. NO/DATE: EU/1/13/846 20130625 |
| 1189916 | C01189916/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: REGADENOSON; REGISTRATION NO/DATE: SWISSMEDIC 66137 28.07.2017 |
| 1893196 | 169 50005-2013 | Slovakia | ⤷ Try a Trial | PRODUCT NAME: ENZALUTAMID; REGISTRATION NO/DATE: EU/1/13/846 20130621 |
| 2428508 | 2090007-2 | Sweden | ⤷ Try a Trial | PRODUCT NAME: GILTERITINIB OR A SALT THEROF; REG. NO/DATE: EU/1/19/1399 20191028 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.