Covis Company Profile
✉ Email this page to a colleague
What is the competitive landscape for COVIS, and what generic alternatives to COVIS drugs are available?
COVIS has eleven approved drugs.
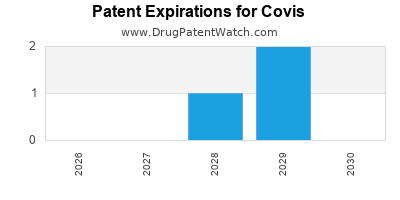
There are five US patents protecting COVIS drugs.
There are one hundred and forty-four patent family members on COVIS drugs in forty-six countries and twenty-seven supplementary protection certificates in fourteen countries.
Drugs and US Patents for Covis
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Covis | ZETONNA | ciclesonide | AEROSOL, METERED;NASAL | 202129-001 | Jan 20, 2012 | RX | Yes | Yes | 8,371,292 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Covis | PRILOSEC | omeprazole magnesium | FOR SUSPENSION, DELAYED RELEASE;ORAL | 022056-001 | Mar 20, 2008 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Covis | DUAKLIR PRESSAIR | aclidinium bromide; formoterol fumarate | POWDER, METERED;INHALATION | 210595-001 | Mar 29, 2019 | RX | Yes | Yes | 8,051,851 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Covis | SULAR | nisoldipine | TABLET, EXTENDED RELEASE;ORAL | 020356-002 | Feb 2, 1995 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Covis | ALVESCO | ciclesonide | AEROSOL, METERED;INHALATION | 021658-002 | Jan 10, 2008 | RX | Yes | Yes | 8,371,292 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Covis | DUAKLIR PRESSAIR | aclidinium bromide; formoterol fumarate | POWDER, METERED;INHALATION | 210595-001 | Mar 29, 2019 | RX | Yes | Yes | 10,085,974 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Covis | OMNARIS | ciclesonide | SPRAY, METERED;NASAL | 022004-001 | Oct 20, 2006 | RX | Yes | Yes | 8,371,292 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Covis
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Covis | ALTOPREV | lovastatin | TABLET, EXTENDED RELEASE;ORAL | 021316-001 | Jun 26, 2002 | 6,485,748 | ⤷ Try a Trial |
| Covis | ZETONNA | ciclesonide | AEROSOL, METERED;NASAL | 202129-001 | Jan 20, 2012 | 6,120,752 | ⤷ Try a Trial |
| Covis | DUAKLIR PRESSAIR | aclidinium bromide; formoterol fumarate | POWDER, METERED;INHALATION | 210595-001 | Mar 29, 2019 | 7,078,412 | ⤷ Try a Trial |
| Covis | SULAR | nisoldipine | TABLET, EXTENDED RELEASE;ORAL | 020356-008 | Jan 2, 2008 | 5,626,874 | ⤷ Try a Trial |
| Covis | ALVESCO | ciclesonide | AEROSOL, METERED;INHALATION | 021658-002 | Jan 10, 2008 | 6,036,942 | ⤷ Try a Trial |
| Covis | DUAKLIR PRESSAIR | aclidinium bromide; formoterol fumarate | POWDER, METERED;INHALATION | 210595-001 | Mar 29, 2019 | 9,056,100 | ⤷ Try a Trial |
| Covis | PRILOSEC | omeprazole magnesium | FOR SUSPENSION, DELAYED RELEASE;ORAL | 022056-002 | Mar 20, 2008 | 6,428,810 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for COVIS drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 40 mg | ➤ Subscribe | 2007-06-11 |
| ➤ Subscribe | Nasal Spray | 250 mcg | ➤ Subscribe | 2012-02-13 |
| ➤ Subscribe | Extended-release Tablets | 25.5 mg and 34 mg | ➤ Subscribe | 2008-11-28 |
| ➤ Subscribe | Extended-release Tablets | 8.5 mg and 17 mg | ➤ Subscribe | 2009-03-02 |
| ➤ Subscribe | Injection | 30 mg/mL, 17 mL single-use vials | ➤ Subscribe | 2015-12-04 |
| ➤ Subscribe | Extended-release Tablets | 20 mg and 30 mg | ➤ Subscribe | 2007-11-07 |
International Patents for Covis Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| South Korea | 20180125055 | ⤷ Try a Trial |
| Poland | 357160 | ⤷ Try a Trial |
| Russian Federation | 2608713 | ⤷ Try a Trial |
| Russian Federation | 2007105156 | ⤷ Try a Trial |
| Estonia | 200200017 | ⤷ Try a Trial |
| Australia | 2005202144 | ⤷ Try a Trial |
| Montenegro | 02286 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Covis Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1200431 | CA 2013 00002 | Denmark | ⤷ Try a Trial | |
| 1200431 | PA2013001 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: ACLIDINII BROMIDUM; REGISTRATION NO/DATE: EU/1/12/778/001 - EU/1/12/778/003, 2012 07 20 EU/1/12/781/001 - EU/1/12/781/003 20120720 |
| 1200431 | C01200431/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: ACLIDINIUM; REGISTRATION NO/DATE: SWISSMEDIC 62590 25.04.2013 |
| 1411900 | 2011C/016 | Belgium | ⤷ Try a Trial | PRODUCT NAME: NAPROXENE ET ESOMEPRAZOLE (SOUS LA FORME D'ESOMEPRAZOLE MAGNESIUM TRIHYDRATE); AUTHORISATION NUMBER AND DATE: BE382505 20101214 |
| 0124495 | SPC/GB01/006 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ESOMEPRAZOLE AS MAGNESIUM TRIHYDRATE; REGISTERED: SE 15945 20000310; SE 15946 20000310; UK PL 17901/0068-0069 20000727 |
| 1200431 | 1390002-2 | Sweden | ⤷ Try a Trial | PRODUCT NAME: AKLIDINIUMSALT MED FARMACEUTISKT ACCEPTABEL ANJON AV EN- ELLER FLERVAERD SYRA, SPECIELLT AKLIDINIUMBROMID; REG. NO/DATE: EU/1/12/778/001 20120720 |
| 1411900 | SPC/GB11/015 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: NAPROXEN AND ESOMEPRAZOLE; REGISTERED: UK PL 17901/0263-0001 20101105 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |