Galderma Labs Lp Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GALDERMA LABS LP, and what generic alternatives to GALDERMA LABS LP drugs are available?
GALDERMA LABS LP has twenty-six approved drugs.
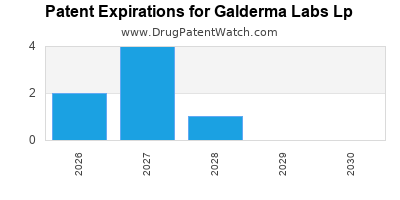
There are fifty-eight US patents protecting GALDERMA LABS LP drugs.
There are four hundred and five patent family members on GALDERMA LABS LP drugs in thirty-seven countries and one hundred and seventy-eight supplementary protection certificates in eighteen countries.
Summary for Galderma Labs Lp
| International Patents: | 405 |
| US Patents: | 58 |
| Tradenames: | 18 |
| Ingredients: | 16 |
| NDAs: | 26 |
Drugs and US Patents for Galderma Labs Lp
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | MIRVASO | brimonidine tartrate | GEL;TOPICAL | 204708-001 | Aug 23, 2013 | AB | RX | Yes | Yes | 8,163,725 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Galderma Labs Lp | EPIDUO | adapalene; benzoyl peroxide | GEL;TOPICAL | 022320-001 | Dec 8, 2008 | AB | RX | Yes | Yes | 7,964,202 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Galderma Labs Lp | DIFFERIN | adapalene | LOTION;TOPICAL | 022502-001 | Mar 17, 2010 | RX | Yes | Yes | 8,435,502 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Galderma Labs Lp
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | METVIXIA | methyl aminolevulinate hydrochloride | CREAM;TOPICAL | 021415-001 | Jul 27, 2004 | 6,034,267 | ⤷ Try a Trial |
| Galderma Labs Lp | EPIDUO | adapalene; benzoyl peroxide | GEL;TOPICAL | 022320-001 | Dec 8, 2008 | 8,105,618 | ⤷ Try a Trial |
| Galderma Labs Lp | DIFFERIN | adapalene | SOLUTION;TOPICAL | 020338-001 | May 31, 1996 | RE34440 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GALDERMA LABS LP drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Delayed-release Capsules | 40 mg | ➤ Subscribe | 2008-12-11 |
| ➤ Subscribe | Cream | 1% | ➤ Subscribe | 2016-12-30 |
| ➤ Subscribe | Topical Gel | 1% | ➤ Subscribe | 2008-10-21 |
| ➤ Subscribe | Lotion | 0.05% | ➤ Subscribe | 2006-03-27 |
| ➤ Subscribe | Topical Shampoo | 0.05% | ➤ Subscribe | 2008-01-09 |
| ➤ Subscribe | Topical Gel | 0.30% | ➤ Subscribe | 2009-09-15 |
| ➤ Subscribe | Gel | 0.1%/2.5% | ➤ Subscribe | 2011-12-30 |
| ➤ Subscribe | Topical Gel | 0.33% | ➤ Subscribe | 2014-12-15 |
| ➤ Subscribe | Spray | 0.05% | ➤ Subscribe | 2008-09-29 |
International Patents for Galderma Labs Lp Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Norway | 333468 | ⤷ Try a Trial |
| Russian Federation | 2320327 | ⤷ Try a Trial |
| Hong Kong | 1223841 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Galderma Labs Lp Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1631293 | 92462 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE ET SES SELS PHARMACEUTIQUES POUR L UTILISATION COMME MEDICAMENT POUR LE TRAITEMENT DES ROUGEURS INDUITES PAR LA ROSACEA.FIRST REGISTRATION: 20140225 |
| 0306228 | SPC/GB01/002 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: 5-(4-(2-(N-METHYL-N-(2-PYRIDYL)AMINO)ETHOXY) BENZYL)-2,4-THIAZOLIDINEDIONE, "ROSIGLITAZONE" ITS TAUTOMERS, PHARMACEUTICALLY ACCEPTABLE SALTS, SUCH AS THE MALEATE, AND PHARMACEUTICALLY ACCEPTABLE SOLVATES.; REGISTERED: CH 55176 02 19990929; CH 55176 03 19990929; CH 55176 04 19990929; UK EU/1/00/137/001 20000711; UK EU/1/00/137/002 20000711; UK EU/1/00/137/003 20000711; UK EU/1/00/137/004 20000711; UK EU/1/00/137/005 20000711; UK EU/1/00/137/006 20000711; UK EU/1/00/137/007 20000711; UK EU/1/00/137/008 20000711; UK EU/1/00/137/009 20000711; UK EU/1/00/137/010 20000711; UK EU/1/00/137/011 20000711; UK EU/1/00/137/012 20000711 |
| 1831149 | CR 2020 00027 | Denmark | ⤷ Try a Trial | PRODUCT NAME: TRIFAROTEN, EVENTUELT I FORM AF ET SALT DERAF; NAT. REG. NO/DATE: 61878 20200120; FIRST REG. NO/DATE: UK PL 10590/0071 20200113 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.