Boehringer Ingelheim Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BOEHRINGER INGELHEIM, and when can generic versions of BOEHRINGER INGELHEIM drugs launch?
BOEHRINGER INGELHEIM has forty-eight approved drugs.
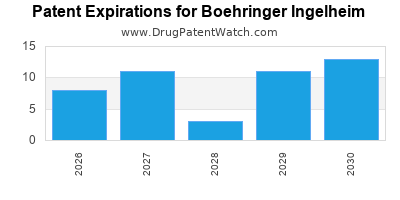
There are sixty-nine US patents protecting BOEHRINGER INGELHEIM drugs.
There are one thousand seven hundred and eighty-two patent family members on BOEHRINGER INGELHEIM drugs in sixty-three countries and two hundred and seventeen supplementary protection certificates in nineteen countries.
Summary for Boehringer Ingelheim
| International Patents: | 1782 |
| US Patents: | 69 |
| Tradenames: | 37 |
| Ingredients: | 29 |
| NDAs: | 48 |
| Patent Litigation for Boehringer Ingelheim: | See patent lawsuits for Boehringer Ingelheim |
| PTAB Cases with Boehringer Ingelheim as petitioner: | See PTAB cases with Boehringer Ingelheim as petitioner |
Drugs and US Patents for Boehringer Ingelheim
Expired US Patents for Boehringer Ingelheim
Paragraph IV (Patent) Challenges for BOEHRINGER INGELHEIM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 80 mg/25 mg | ➤ Subscribe | 2009-02-27 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2018-10-15 |
| ➤ Subscribe | Tablets | 0.25 mg | ➤ Subscribe | 2005-05-27 |
| ➤ Subscribe | Capsules | eq. to 110 mg base | ➤ Subscribe | 2015-12-15 |
| ➤ Subscribe | Oral Suspension | 7.5 mg/5 mL | ➤ Subscribe | 2009-12-17 |
| ➤ Subscribe | Extended-releaseTablets | 2.5 mg/1000 mg 5 mg/1000 mg | ➤ Subscribe | 2018-03-28 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Extended-release Tablets | 2.25 mg and 3.75 mg | ➤ Subscribe | 2011-07-26 |
| ➤ Subscribe | Extended-release Tablets | 400 mg | ➤ Subscribe | 2013-06-21 |
| ➤ Subscribe | Tablets | 10 mg/5 mg and 25 mg/5 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 2.5 mg/500 mg, 2.5 mg/850 mg, 2.5 mg/1000 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Tablets | 80 mg/12.5 mg and 40 mg/12.5 mg | ➤ Subscribe | 2008-12-31 |
| ➤ Subscribe | Extended-release Capsules | 25 mg and 200 mg | ➤ Subscribe | 2007-02-01 |
| ➤ Subscribe | Tablets | 0.125 mg, 0.5 mg, 1 mg and 1.5 mg | ➤ Subscribe | 2005-06-24 |
| ➤ Subscribe | Capsules | eq. to 75 mg base and 150 mg base | ➤ Subscribe | 2014-10-20 |
| ➤ Subscribe | Tablets | 0.75 mg | ➤ Subscribe | 2008-07-31 |
| ➤ Subscribe | Inhalation Powder Capsules | 18 mcg | ➤ Subscribe | 2018-05-11 |
| ➤ Subscribe | Tablets | 5 mg/500 mg5 mg/1000 mg12.5 mg/500 mg12.5 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Extended-release Tablets | 0.375 mg, 0.75 mg, 1.5 mg, 3 mg and 4.5 mg | ➤ Subscribe | 2010-06-01 |
| ➤ Subscribe | Extended-release Tablets | 5 mg/1000 mg10 mg/1000 mg12.5 mg/1000 mg25 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 30 mg and 40 mg | ➤ Subscribe | 2017-07-12 |
| ➤ Subscribe | Tablets | 10 mg and 25 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 40 mg and 80 mg | ➤ Subscribe | 2006-12-26 |
International Patents for Boehringer Ingelheim Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Colombia | 6571889 | ⤷ Try a Trial |
| South Korea | 101781787 | ⤷ Try a Trial |
| Canada | 2749579 | ⤷ Try a Trial |
| Brazil | 112012018021 | ⤷ Try a Trial |
| Japan | 5668024 | ⤷ Try a Trial |
| South Korea | 102051281 | ⤷ Try a Trial |
| Uruguay | 30321 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Boehringer Ingelheim Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0418716 | 300084 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: ZOUTEN VAN TRIOTROPIUM, IN HET BIJZONDER TIOTROPIUM BROMIDE; REGISTRATION NO/DATE: RVG 26191 20011009 |
| 0503785 | CA 2011 00026 | Denmark | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION OF OLMESARTAN MEDOXOMIL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, AND AMLODIPINE BESYLATE AND HYDROCHLOROTHIAZIDE; NAT. REG. NO/DATE: 46260-46269 (DK) 20110323; FIRST REG. NO/DATE: DE 79810.00.00 20101216 |
| 0480717 | 98C0025 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LOSARTAN POTASSIUM; HYDROCHLOROTHIAZIDE; NAT. REGISTRATION NO/DATE: NL 20 037 19950215; FIRST REGISTRATION: FR - NL 20 037 19950215 |
| 1562603 | CR 2014 00014 | Denmark | ⤷ Try a Trial | PRODUCT NAME: OLODATEROL, OPTISKE ISOMERER DERAF, BLANDINGER AF ISOMERER DERAF, SYREADDITIONSSALTE DERAF MED FARMAKOLGISK HARMLOESE SYRER, SAVEL SOM SOLVATER OG/ELLER HYDRATER DERAF, SAERLIGT OLODATEROL OG OLODATEROL HYDROCHLORID; NAT. REG. NO/DATE: 50975 20131014; FIRST REG. NO/DATE: MA 211/00401 20130918 |
| 1830843 | C300747 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: NINTEDANIB, OF EEN TAUTOMEER,; REGISTRATION NO/DATE: EU/1/14/979 20150115 |
| 0429987 | 99C0019 | France | ⤷ Try a Trial | PRODUCT NAME: NEVIRAPINE; NAT. REGISTRATION NO/DATE: EU/1/97/055/001 19980205; FIRST REGUSTRATION: CH/LI 54 393 19971223 |
| 1730131 | 122014000099 | Germany | ⤷ Try a Trial | PRODUCT NAME: EMPAGLIFLOZIN UND DESSEN SALZE, INSBESONDERE EMPAGLIFLOZIN; REGISTRATION NO/DATE: EU/1/14/930 20140522 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.