Glaxosmithkline Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GLAXOSMITHKLINE
GLAXOSMITHKLINE has one hundred and fifty-two approved drugs.
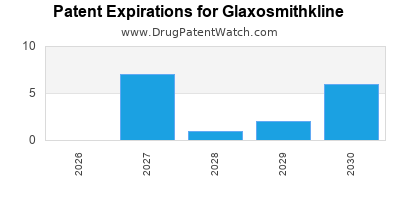
There are thirty-seven US patents protecting GLAXOSMITHKLINE drugs.
There are eight hundred and five patent family members on GLAXOSMITHKLINE drugs in sixty-two countries and one hundred and eighty-four supplementary protection certificates in twenty-one countries.
Summary for Glaxosmithkline
| International Patents: | 805 |
| US Patents: | 37 |
| Tradenames: | 107 |
| Ingredients: | 93 |
| NDAs: | 152 |
| Drug Master File Entries: | 8 |
| Patent Litigation for Glaxosmithkline: | See patent lawsuits for Glaxosmithkline |
| PTAB Cases with Glaxosmithkline as petitioner: | See PTAB cases with Glaxosmithkline as petitioner |
Drugs and US Patents for Glaxosmithkline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glaxosmithkline | IMITREX | sumatriptan succinate | TABLET;ORAL | 020132-001 | Jun 1, 1995 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline | SYNCURINE | decamethonium bromide | INJECTABLE;INJECTION | 006931-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | IMITREX STATDOSE | sumatriptan succinate | INJECTABLE;SUBCUTANEOUS | 020080-003 | Dec 23, 1996 | AB | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | ||||
| Glaxosmithkline | FASTIN | phentermine hydrochloride | CAPSULE;ORAL | 017352-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| Glaxosmithkline | ACTIFED | pseudoephedrine hydrochloride; triprolidine hydrochloride | CAPSULE, EXTENDED RELEASE;ORAL | 018996-001 | Jun 17, 1985 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Glaxosmithkline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Glaxosmithkline | WELLBUTRIN SR | bupropion hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020358-003 | Oct 4, 1996 | RE33994 | ⤷ Start Trial |
| Glaxosmithkline | RELENZA | zanamivir | POWDER;INHALATION | 021036-001 | Jul 26, 1999 | 4,811,731 | ⤷ Start Trial |
| Glaxosmithkline | WELLBUTRIN SR | bupropion hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020358-003 | Oct 4, 1996 | 5,427,798 | ⤷ Start Trial |
| Glaxosmithkline | ARNUITY ELLIPTA | fluticasone furoate | POWDER;INHALATION | 205625-003 | May 17, 2018 | 7,629,335 | ⤷ Start Trial |
| Glaxosmithkline | WELLBUTRIN | bupropion hydrochloride | TABLET;ORAL | 018644-001 | Dec 30, 1985 | 4,507,323 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GLAXOSMITHKLINE drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Extended-release Tablets | 6 mg | ➤ Subscribe | 2009-07-22 |
| ➤ Subscribe | Tablets | 150 mg | ➤ Subscribe | 2007-10-30 |
| ➤ Subscribe | Orally Disintegrating Tablets | 25 mg, 50 mg, 100 mg, and 200 mg | ➤ Subscribe | 2009-12-21 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL (prefilled syringes) | ➤ Subscribe | 2006-05-09 |
| ➤ Subscribe | Tablets | 3 mg, 4 mg and 5 mg | ➤ Subscribe | 2005-02-04 |
| ➤ Subscribe | Tablets | 250 mg/100 mg | ➤ Subscribe | 2009-04-03 |
| ➤ Subscribe | Extended-release Capsules | 325 mg | ➤ Subscribe | 2006-11-07 |
| ➤ Subscribe | Oral Suspension | 750 mg/5 mL | ➤ Subscribe | 2009-10-20 |
| ➤ Subscribe | Extended-release Tablets | 4 mg | ➤ Subscribe | 2008-10-31 |
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Extended-release Tablets | 2 mg | ➤ Subscribe | 2008-10-14 |
| ➤ Subscribe | Extended-release Tablets | 3 mg | ➤ Subscribe | 2009-01-08 |
| ➤ Subscribe | Injection | 6 mg/0.5 mL, 0.5 mL vials | ➤ Subscribe | 2004-10-25 |
| ➤ Subscribe | Tablets | 0.25 mg, 0.5 mg, 1 mg and 2 mg | ➤ Subscribe | 2004-12-22 |
| ➤ Subscribe | Tablets | 100 mg | ➤ Subscribe | 2007-10-31 |
| ➤ Subscribe | Tablets | 62.5 mg/25 mg | ➤ Subscribe | 2010-09-14 |
| ➤ Subscribe | Extended-release Capsules | 225 mg and 425 mg | ➤ Subscribe | 2006-10-11 |
| ➤ Subscribe | Extended-release Tablets | 25 mg, 50 mg, 100 mg, 200 mg, 250 mg, and 300 mg | ➤ Subscribe | 2014-02-12 |
| ➤ Subscribe | Extended-release Tablets | 8 mg | ➤ Subscribe | 2008-11-03 |
| ➤ Subscribe | Extended-release Tablets | 12 mg | ➤ Subscribe | 2009-02-05 |
International Patents for Glaxosmithkline Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| South Korea | 20090097191 | ⤷ Start Trial |
| Hungary | E051360 | ⤷ Start Trial |
| Norway | 2506844 | ⤷ Start Trial |
| Slovenia | 2336120 | ⤷ Start Trial |
| Brazil | 122020010759 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Glaxosmithkline Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2924034 | 300981 | Netherlands | ⤷ Start Trial | PRODUCT NAME: DORAVIRINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT IN COMBINATIE MET LAMIVUDINE, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN COMBINATIE MET TENOFOVIR OF EEN ESTER DAARVAN, IN HET BIJZONDER EEN DISOPROXIL ESTER, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT, IN HET BIJZONDER EEN FUMARAAT ZOUT; REGISTRATION NO/DATE: EU/1/18/1333/001-002 20181126 |
| 3494972 | 301277 | Netherlands | ⤷ Start Trial | PRODUCT NAME: DOLUTEGRAVIR OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT, WAARONDER DOLUTEGRAVIRNATRIUM, EN LAMIVUDINE; REGISTRATION NO/DATE: EU/1/19/1370 20190703 |
| 1740177 | CA 2014 00052 | Denmark | ⤷ Start Trial | PRODUCT NAME: UMECLIDINIUMBROMID; REG. NO/DATE: EU/1/14/922/001-003 20140428 |
| 0299602 | SPC/GB96/040 | United Kingdom | ⤷ Start Trial | PRODUCT NAME: ROPINIROLE, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR THE HYDROCHLORIDE SALT; REGISTERED: UK 10592/0085 19960702 |
| 0565634 | 06C0030 | France | ⤷ Start Trial | PRODUCT NAME: EPROSARTAN MESYLATE; HYDROCHLOROTHIAZIDE; NAT. REGISTRATION NO/DATE: NL 32075 20060623; FIRST REGISTRATION: LI - 55783 01 20020607 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.