Lupin Company Profile
✉ Email this page to a colleague
What is the competitive landscape for LUPIN, and when can generic versions of LUPIN drugs launch?
LUPIN has two hundred and thirty-one approved drugs.
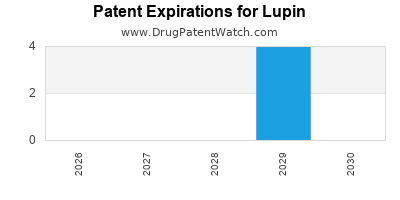
There are nineteen US patents protecting LUPIN drugs. There are twenty-two tentative approvals on LUPIN drugs.
There are ninety-nine patent family members on LUPIN drugs in twenty-four countries and six hundred and seventy-eight supplementary protection certificates in seventeen countries.
Summary for Lupin
| International Patents: | 99 |
| US Patents: | 19 |
| Tradenames: | 189 |
| Ingredients: | 169 |
| NDAs: | 231 |
| Patent Litigation for Lupin: | See patent lawsuits for Lupin |
| PTAB Cases with Lupin as petitioner: | See PTAB cases with Lupin as petitioner |
Drugs and US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin Ltd | MELOXICAM | meloxicam | CAPSULE;ORAL | 209487-001 | Jun 1, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Pharms | SUPRAX | cefixime | TABLET;ORAL | 065130-001 | Feb 12, 2004 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin Ltd | DECITABINE | decitabine | INJECTABLE;INTRAVENOUS | 210756-001 | Nov 9, 2018 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | TADALAFIL | tadalafil | TABLET;ORAL | 210567-002 | Mar 26, 2019 | AB1 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | QUETIAPINE FUMARATE | quetiapine fumarate | TABLET, EXTENDED RELEASE;ORAL | 204203-002 | May 17, 2017 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Lupin Ltd | SUPRAX | cefixime | TABLET, CHEWABLE;ORAL | 065380-002 | Oct 25, 2010 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Lupin | CEFPROZIL | cefprozil | TABLET;ORAL | 065276-002 | Dec 8, 2005 | AB | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Lupin
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,472,563 | ⤷ Try a Trial |
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,068,833 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,844,002 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,760,090 | ⤷ Try a Trial |
| Lupin | XOPENEX HFA | levalbuterol tartrate | AEROSOL, METERED;INHALATION | 021730-001 | Mar 11, 2005 | 5,439,670 | ⤷ Try a Trial |
| Lupin | BROVANA | arformoterol tartrate | SOLUTION;INHALATION | 021912-001 | Oct 6, 2006 | 6,589,508 | ⤷ Try a Trial |
| Lupin | ANTARA (MICRONIZED) | fenofibrate | CAPSULE;ORAL | 021695-001 | Nov 30, 2004 | 7,863,331 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for LUPIN drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | for Injection | 200 mcg/vial | ➤ Subscribe | 2015-05-01 |
| ➤ Subscribe | for Oral Suspension | 500 mg/5 mL | ➤ Subscribe | 2014-07-22 |
| ➤ Subscribe | for Injection | 100 mcg/vial and 500 mcg/vial | ➤ Subscribe | 2015-04-14 |
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
| ➤ Subscribe | Inhalation Solution | Eq. 0.015 mg base/2 mL | ➤ Subscribe | 2009-10-01 |
International Patents for Lupin Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Norway | 331649 | ⤷ Try a Trial |
| Japan | 2012072160 | ⤷ Try a Trial |
| Israel | 168739 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2004052835 | ⤷ Try a Trial |
| European Patent Office | 2748205 | ⤷ Try a Trial |
| European Patent Office | 1572622 | ⤷ Try a Trial |
| World Intellectual Property Organization (WIPO) | 2013130666 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Lupin Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0542411 | 09C0031 | France | ⤷ Try a Trial | PRODUCT NAME: PRASUGREL; NAT. REGISTRATION NO/DATE: EU/1/08/503/001-014 20090225; FIRST REGISTRATION: EU/1/08/503/001/-014 20090225 |
| 1467728 | C01467728/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: SACUBRITIL UND VALSARTAN; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 65673 17.09.2015 |
| 0230742 | 2001C/040 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MONOHYDRATE DU CHLORHYDRATE DE N,N-DIMETHYL-1-4-CHLOROPHENYL)CYCLOBUTYL-3-METHYLBUTYLAMINE ET COMPOSITIONS PHARMACEUTIQUES LE CONTENANT; NAT. REGISTRTION NO/DATE: 479 IS 52 F 5 20010425; FIRST REGISTRATION: CH 54770 19990113 |
| 2316456 | LUC00054 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: NALTREXONE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR NALTREXONE HYDROCHLORIDE, AND BUPROPION OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, IN PARTICULAR BUPROPION HYDROCHLORIDE; AUTHORISATION NUMBER AND DATE: EU/1/14/988 20150330 |
| 1663240 | 15C0073 | France | ⤷ Try a Trial | PRODUCT NAME: COMBINAISON DE RILPIVIRINE OU SES FORMES THERAPEUTIQUEMENT EQUIVALENTES PROTEGEES PAR LE BREVET DE BASE TELLES QUE LES SELS PHARMACEUTIQUEMENT ACCEPTABLES DE RILPIVIRINE Y COMPRIS LE SEL D'ACIDE CHLORHYDRIQUE DE RILPIVIRINE DE TENOFOVIR EN PARTICULIER LE TENOFOVIR DISOPROXIL FUMARATE AINSI QUE D'EMTRICITABINE; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 0771217 | CA 2006 00038 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL (SOM BETA-CYCLODEXTRIN-CLATHRAT) OG DROSPIRENON; NAT. REG. NO/DATE: 38687 20060627; FIRST REG. NO/DATE: EU RVG 31781 20050804 |
| 2441753 | SPC/GB17/036 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ALECTINIB (9-ETHYL-6,6-DIMETHYL-8-(4-MORPHOLIN-4-YL-PIPERIDIN-1-YL)-11-OXO-6,11-DIHYDRO-5H-BENZO(B)CARBAZOLE-3-CARBONITRILE) OR A SALT OR SOLVATE THEREOF; REGISTERED: UK EU/1/16/1169 (NI) 20170220; UK PLGB 00031/0843 20170220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.