Teva Branded Pharm Company Profile
✉ Email this page to a colleague
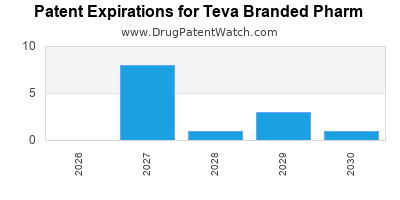
What is the competitive landscape for TEVA BRANDED PHARM, and when can generic versions of TEVA BRANDED PHARM drugs launch?
TEVA BRANDED PHARM has thirty-three approved drugs.
There are fifty-nine US patents protecting TEVA BRANDED PHARM drugs.
There are six hundred and eighty patent family members on TEVA BRANDED PHARM drugs in forty-one countries and thirty-nine supplementary protection certificates in fourteen countries.
Summary for Teva Branded Pharm
| International Patents: | 680 |
| US Patents: | 59 |
| Tradenames: | 33 |
| Ingredients: | 19 |
| NDAs: | 33 |
| Patent Litigation for Teva Branded Pharm: | See patent lawsuits for Teva Branded Pharm |
Drugs and US Patents for Teva Branded Pharm
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Branded Pharm | PROAIR DIGIHALER | albuterol sulfate | POWDER, METERED;INHALATION | 205636-002 | Dec 21, 2018 | RX | Yes | No | 8,978,966 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-002 | Apr 3, 2017 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-001 | Apr 3, 2017 | RX | Yes | No | 11,564,917*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-003 | Apr 3, 2017 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Teva Branded Pharm | VANTRELA ER | hydrocodone bitartrate | TABLET, EXTENDED RELEASE;ORAL | 207975-001 | Jan 17, 2017 | DISCN | Yes | No | 9,216,176 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Teva Branded Pharm | SEASONIQUE | ethinyl estradiol; levonorgestrel | TABLET;ORAL | 021840-001 | May 25, 2006 | AB | RX | Yes | Yes | 7,855,190 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Teva Branded Pharm | AUSTEDO | deutetrabenazine | TABLET;ORAL | 208082-002 | Apr 3, 2017 | RX | Yes | No | 11,179,386*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Teva Branded Pharm
Paragraph IV (Patent) Challenges for TEVA BRANDED PHARM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 1 mg/0.02 mg and 75 mg | ➤ Subscribe | 2006-04-17 |
| ➤ Subscribe | Tablets | 0.15 mg/0.02 mg, 0.15 mg/0.025 mg, 0.15 mg/0.03 mg and 0.01 mg | ➤ Subscribe | 2013-07-10 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg/0.01 mg | ➤ Subscribe | 2008-01-22 |
| ➤ Subscribe | Tablets | 0.1 mg/0.02 mg and 0.01 mg | ➤ Subscribe | 2009-11-16 |
| ➤ Subscribe | Tablets | 0.15 mg/0.03 mg | ➤ Subscribe | 2004-03-29 |
International Patents for Teva Branded Pharm Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2022287570 | ⤷ Try a Trial |
| Canada | 2814212 | ⤷ Try a Trial |
| European Patent Office | 2305230 | ⤷ Try a Trial |
| Croatia | P20150452 | ⤷ Try a Trial |
| Slovenia | 2692341 | ⤷ Try a Trial |
| Eurasian Patent Organization | 201991218 | ⤷ Try a Trial |
| Eurasian Patent Organization | 023795 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Teva Branded Pharm Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1389098 | 2013C/054 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LOXAPINE; AUTHORISATION NUMBER AND DATE: EU/1/13/823/001 20130225 |
| 1453521 | 93156 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: LEVONORGESTREL ET ETHINYLESTRADIOL; FIRST REGISTRATION DATE: 20150211 |
| 1214076 | C01214076/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: DROSPIRENONE + ETHINYLESTRADIOL; REGISTRATION NUMBER/DATE: SWISSMEDIC 57946 13.06.2008 |
| 0502314 | C300095 | Netherlands | ⤷ Try a Trial | PRODCUT NAME: TELMISARTAN, DESGEWENST IN DE VORM VAN EEN FYSIOLOGISCH VERDRAAGBAAR ZOUT, EN HYDROCHLOROTHIAZIDE; REGISTRATION NO/DATE: EU/1/02/213/001-010 20020419 |
| 1380301 | CA 2009 00017 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ETHINYLESTRADIOL (SOM BETADEXCLATHRAT) OG DROSPIRENON; NAT. REG. NO/DATE: 42417 (DK) 20080619; FIRST REG. NO/DATE: NL 33842 20070629 |
| 0565634 | 06C0030 | France | ⤷ Try a Trial | PRODUCT NAME: EPROSARTAN MESYLATE; HYDROCHLOROTHIAZIDE; NAT. REGISTRATION NO/DATE: NL 32075 20060623; FIRST REGISTRATION: LI - 55783 01 20020607 |
| 1389098 | C300609 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: LOXAPINE; REGISTRATION NO/DATE: EU/1/13/823/001-002 20130220 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.