RIFAMPIN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Rifampin, and what generic alternatives are available?
Rifampin is a drug marketed by Chartwell Molecular, Epic Pharma Llc, Hikma, Lupin Pharms, Avet Lifesciences, Fresenius Kabi Usa, Hikma Pharms, Mylan Labs Ltd, Watson Pharms Teva, and Hikma Intl Pharms. and is included in twelve NDAs.
The generic ingredient in RIFAMPIN is isoniazid; rifampin. There are five drug master file entries for this compound. Additional details are available on the isoniazid; rifampin profile page.
Summary for RIFAMPIN
| US Patents: | 0 |
| Applicants: | 10 |
| NDAs: | 12 |
| Finished Product Suppliers / Packagers: | 14 |
| Raw Ingredient (Bulk) Api Vendors: | 47 |
| Clinical Trials: | 306 |
| Patent Applications: | 4,554 |
| Formulation / Manufacturing: | see details |
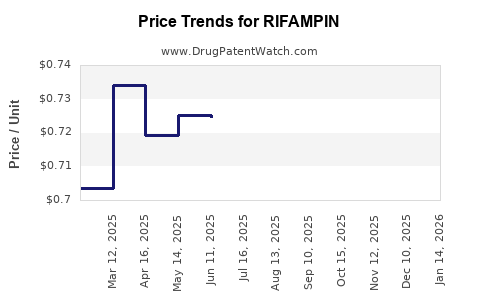
| Drug Prices: | Drug price information for RIFAMPIN |
| DailyMed Link: | RIFAMPIN at DailyMed |
Recent Clinical Trials for RIFAMPIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Assistance Publique Hopitaux De Marseille | Phase 3 |
| Hôpital Necker-Enfants Malades | Phase 3 |
| Institut Pasteur | Phase 3 |
Pharmacology for RIFAMPIN
| Drug Class | Rifamycin Antibacterial |
Medical Subject Heading (MeSH) Categories for RIFAMPIN
US Patents and Regulatory Information for RIFAMPIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Chartwell Molecular | RIFAMPIN | rifampin | CAPSULE;ORAL | 065390-001 | Mar 28, 2008 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lupin Pharms | RIFAMPIN | rifampin | CAPSULE;ORAL | 090034-001 | Aug 21, 2013 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Epic Pharma Llc | RIFAMPIN | rifampin | CAPSULE;ORAL | 064150-001 | May 28, 1997 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Hikma | RIFAMPIN | rifampin | INJECTABLE;INJECTION | 064217-001 | Oct 29, 1999 | AP | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


