CEFUROXIME Drug Patent Profile
✉ Email this page to a colleague
When do Cefuroxime patents expire, and what generic alternatives are available?
Cefuroxime is a drug marketed by B Braun, Sun Pharm Inds Ltd, Alkem Labs Ltd, Anda Repository, Ani Pharms, Apotex, Aurobindo Pharma, Chartwell Rx, Fosun Pharma, Lupin, Ranbaxy Labs Ltd, Acs Dobfar Spa, Fresenius Kabi Usa, Hikma, Hospira Inc, Teva Pharms, Watson Labs Inc, and Samson Medcl. and is included in twenty-six NDAs.
The generic ingredient in CEFUROXIME is cefuroxime axetil. There are sixty-two drug master file entries for this compound. Sixteen suppliers are listed for this compound. Additional details are available on the cefuroxime axetil profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Cefuroxime
A generic version of CEFUROXIME was approved as cefuroxime axetil by APOTEX on October 2nd, 2002.
Summary for CEFUROXIME
| US Patents: | 0 |
| Applicants: | 18 |
| NDAs: | 26 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for CEFUROXIME |
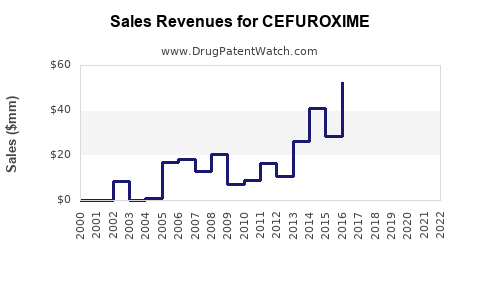
| Drug Sales Revenues: | Drug sales revenues for CEFUROXIME |
| DailyMed Link: | CEFUROXIME at DailyMed |
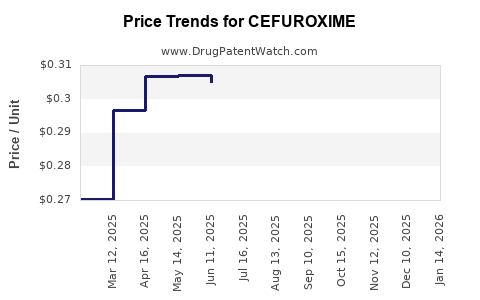
See drug prices for CEFUROXIME
Pharmacology for CEFUROXIME
| Drug Class | Cephalosporin Antibacterial |
US Patents and Regulatory Information for CEFUROXIME
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Watson Labs Inc | CEFUROXIME SODIUM | cefuroxime sodium | INJECTABLE;INJECTION | 064036-001 | Feb 26, 1993 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Fresenius Kabi Usa | CEFUROXIME SODIUM | cefuroxime sodium | INJECTABLE;INJECTION | 065001-002 | May 30, 2001 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Samson Medcl | CEFUROXIME SODIUM IN PLASTIC CONTAINER | cefuroxime sodium | INJECTABLE;INJECTION | 065251-001 | Dec 30, 2009 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Hospira Inc | CEFUROXIME SODIUM | cefuroxime sodium | INJECTABLE;INTRAMUSCULAR, INTRAVENOUS | 065483-001 | Oct 15, 2008 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


