DAKLINZA Drug Patent Profile
✉ Email this page to a colleague
When do Daklinza patents expire, and when can generic versions of Daklinza launch?
Daklinza is a drug marketed by Bristol-myers Squibb and is included in one NDA. There are five patents protecting this drug.
This drug has ninety-three patent family members in thirty-one countries.
The generic ingredient in DAKLINZA is daclatasvir dihydrochloride. There are five drug master file entries for this compound. Additional details are available on the daclatasvir dihydrochloride profile page.
DrugPatentWatch® Generic Entry Outlook for Daklinza
Daklinza was eligible for patent challenges on July 24, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 13, 2031. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
Summary for DAKLINZA
| International Patents: | 93 |
| US Patents: | 5 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 128 |
| Clinical Trials: | 13 |
| Patent Applications: | 584 |
| Drug Prices: | Drug price information for DAKLINZA |
| DailyMed Link: | DAKLINZA at DailyMed |
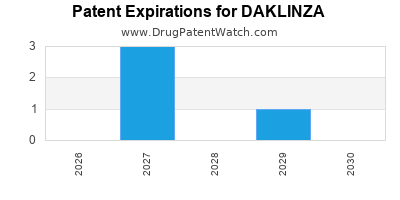

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for DAKLINZA
Generic Entry Date for DAKLINZA*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for DAKLINZA
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Cairo University | Phase 3 |
| Ain Shams University | Phase 3 |
| Tanta University | Phase 2/Phase 3 |
Anatomical Therapeutic Chemical (ATC) Classes for DAKLINZA
US Patents and Regulatory Information for DAKLINZA
DAKLINZA is protected by eight US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of DAKLINZA is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting DAKLINZA
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Crystalline form of methyl ((1S)-1-((25)-2-(5-(4'-(2-((25)-1((2S)-2-((methoxycarbonyl)amino)-3-methy- lbutanoyl)-2-pyrrolidinyl)-1H-imidazol-2-yl)-1-pyrrolidinyl)carbonyl)-2-me- thylpropyl)carbamate dihydrochloride salt
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS WITH DAKLINZA AND AT LEAST ONE ADDITIONAL COMPOUND HAVING ANTI-HCV ACTIVITY
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS WITH DAKLINZA AND AT LEAST ONE ADDITIONAL COMPOUND HAVING ANTI-HCV ACTIVITY
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS WITH DAKLINZA AND AT LEAST ONE ADDITIONAL COMPOUND HAVING ANTI-HCV ACTIVITY
Hepatitis C virus inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Patented Use: METHOD OF INHIBITING HEPATITIS C VIRUS
International Patents for DAKLINZA
When does loss-of-exclusivity occur for DAKLINZA?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 0016
Estimated Expiration: ⤷ Try a Trial
Patent: 4566
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 08284100
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0815142
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 95729
Estimated Expiration: ⤷ Try a Trial
Chile
Patent: 08002354
Estimated Expiration: ⤷ Try a Trial
China
Patent: 1778840
Estimated Expiration: ⤷ Try a Trial
Colombia
Patent: 60327
Estimated Expiration: ⤷ Try a Trial
Eurasian Patent Organization
Patent: 8152
Estimated Expiration: ⤷ Try a Trial
Patent: 1000196
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 83244
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 44089
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 3684
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 44179
Estimated Expiration: ⤷ Try a Trial
Patent: 10535785
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 10001368
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 3148
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 090940
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 1000843
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1508022
Estimated Expiration: ⤷ Try a Trial
Patent: 100042641
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 02791
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 59813
Estimated Expiration: ⤷ Try a Trial
Patent: 0911790
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering DAKLINZA around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 101558059 | Hepatitis c virus inhibitors | ⤷ Try a Trial |
| Lithuania | C2049522 | ⤷ Try a Trial | |
| New Zealand | 583148 | CRYSTALLINE FORM OF METHYL ((1S)-1-(((2S)-2-(5-(4'-(2-((2S)-1-((2S)-2-((METHOXYCARBONYL)AMINO)-3-METHYLBUTANOYL)-2-PYRROLIDINYL)-1H-IMIDAZOL-5-YL)-4-BIPHENYLYL)-1H-IMIDAZOL-2-YL)-1-PYRROLIDINYL)CARBONYL)-2-METHYLPROPYL)CARBAMATE DIHYDROCHLORIDE SALT | ⤷ Try a Trial |
| South Korea | 101508022 | ⤷ Try a Trial | |
| Taiwan | 200911790 | Crystalline form of methyl ((1S)-1-(((2S)-2-(5-(4'-(2-((2S)-1-((2S)-2-((methoxycarbonyl)amino)-3-methylbutanoyl)-2-pyrrolidinyl)-1H-imidazol-5-yl)-4-biphenylyl)-1H-imidazol-2-yl)-1-pyrrolidinyl)carbonyl)-2-methylpropyl)carbamate dihydrochloride salt | ⤷ Try a Trial |
| Canada | 2695729 | FORME CRISTALLINE DE DIHYDROCHLORURE DE METHYL ((1S)-1-(((2S>2-(5-(4'-(2-((2S)-1-((2S)-2-((METHOXYCARBONYL)AMINO)-3-METHYLBUTANOYL)-2-PYRROLIDINYL)-1H-IMIDAZOL-5-YL)-4-BIPHENYLYL)-1H-IMIDAZOL-2-YL)-1-PYRROLIDINYL)CARBONYL)-2-METHYLPROPYL)CARBAMATE (CRYSTALLINE FORM OF METHYL ((1S)-1-(((2S)-2-(5-(4'-(2-((2S)-1-((2S)-2-((METHOXYCARBONYL)AMINO)-3-METHYLBUTANOYL)-2-PYRROLIDINYL)-1H-IMIDAZOL-5-YL)-4-BIPHENYLYL)-1H-IMIDAZOL-2-YL)-1-PYRROLIDINYL)CARBONYL)-2-METHYLPROPYL)CARBAMATE DIHYDROCHLORIDE SALT) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for DAKLINZA
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2049522 | 9/2015 | Austria | ⤷ Try a Trial | PRODUCT NAME: DECLATASVIR; REGISTRATION NO/DATE: EU/1/14/939 (MITTEILUNG) 20140826 |
| 2049522 | 92635 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR ET LEURS SELS PHARMACEUTIQUEMENT ACCEPTABLES , EN PARTICULIER DICHLORHYDRATE DE DACLATASVIR. FIRST REGISTRATION: 20140826 |
| 2049522 | PA2015006,C2049522 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: DAKLATASVIRAS IR JO FARMACINIU POZIURIU PRIIMTINOS DRUSKOS, YPAC DAKLATASVIRO HIDROCHLORIDAS; REGISTRATION NO/DATE: EU/1/14/939/001 - EU/1/14/939/004, 2 00140822 |
| 2049522 | C300713 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR EN FARMACEUTISCH; REGISTRATION NO/DATE: EU/1/14/939/001-004 20150119 |
| 2049522 | SPC/GB15/003 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: DACLATASVIR AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF, ESPECIALLY DACLATASVIR DIHYDROCHLORIDE; REGISTERED: UK EU/1/14/939/001-004 20140826 |
| 2049522 | C20150003 00128 | Estonia | ⤷ Try a Trial | PRODUCT NAME: DAKLATASVIIR;REG NO/DATE: EU/1/14/939 26.08.2014 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


