FLUOXETINE Drug Patent Profile
✉ Email this page to a colleague
When do Fluoxetine patents expire, and what generic alternatives are available?
Fluoxetine is a drug marketed by Sun Pharm Industries, Watson Labs, Barr, Dr Reddys Labs Ltd, Accord Hlthcare, Alembic Pharms Ltd, Ani Pharms, Apnar Pharma Lp, Aurobindo Pharma, Beximco Pharms Usa, Cadila Pharms Ltd, Carlsbad, Cr Double Crane, Granules, Heritage Pharms, Ivax Sub Teva Pharms, Landela Pharm, Marksans Pharma, Micro Labs, Mylan, Mylan Pharms Inc, Natco Pharma Usa, Sandoz, Sciegen Pharms Inc, Specgx Llc, Strides Pharma, Sun Pharm Inds Ltd, Teva, Teva Pharms Usa, Actavis Mid Atlantic, Akorn, Aptapharma Inc, Chartwell Molecular, Lannett Co Inc, Nostrum Labs Inc, Novitium Pharma, Pharm Assoc, Pharmobedient Cnsltg, Alembic, Aurobindo Pharma Ltd, Dr Reddys, Fosun Pharma, G And W Labs Inc, Inventia Hlthcare, Lupin Ltd, Par Form, Par Pharm Inc, Rising, Slate Run Pharma, Taro, Torrent, Twi Pharms, and Upsher Smith Labs. and is included in sixty-seven NDAs.
The generic ingredient in FLUOXETINE is fluoxetine hydrochloride. There are twenty-seven drug master file entries for this compound. Sixty-nine suppliers are listed for this compound. Additional details are available on the fluoxetine hydrochloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Fluoxetine
A generic version of FLUOXETINE was approved as fluoxetine hydrochloride by APNAR PHARMA LP on August 2nd, 2001.
Summary for FLUOXETINE
| US Patents: | 0 |
| Applicants: | 53 |
| NDAs: | 67 |
| Raw Ingredient (Bulk) Api Vendors: | 86 |
| Clinical Trials: | 294 |
| Patent Applications: | 4,421 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for FLUOXETINE |
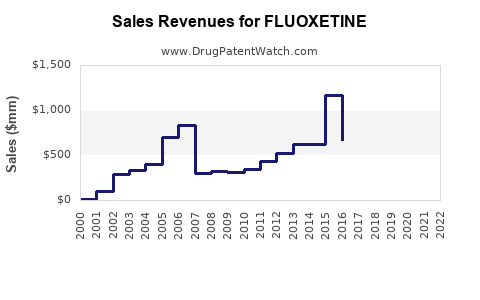
| Drug Sales Revenues: | Drug sales revenues for FLUOXETINE |
| DailyMed Link: | FLUOXETINE at DailyMed |
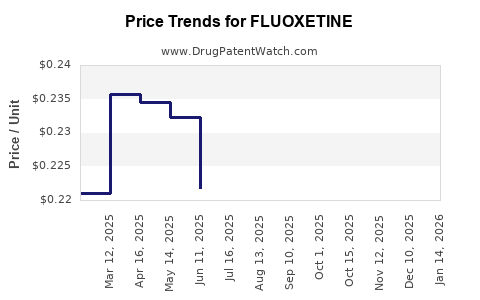
See drug prices for FLUOXETINE
Recent Clinical Trials for FLUOXETINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| First Affiliated Hospital Xi'an Jiaotong University | N/A |
| Chen Qian | Early Phase 1 |
| First Affiliated Hospital of Chongqing Medical University | Phase 1/Phase 2 |
Anatomical Therapeutic Chemical (ATC) Classes for FLUOXETINE
US Patents and Regulatory Information for FLUOXETINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Pharms Usa | FLUOXETINE HYDROCHLORIDE | fluoxetine hydrochloride | CAPSULE;ORAL | 076001-002 | Jan 29, 2002 | AB1 | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Lupin Ltd | FLUOXETINE HYDROCHLORIDE | fluoxetine hydrochloride | TABLET;ORAL | 211632-001 | Feb 8, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Ivax Sub Teva Pharms | FLUOXETINE HYDROCHLORIDE | fluoxetine hydrochloride | CAPSULE;ORAL | 075245-003 | Sep 28, 2004 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


