MEPROBAMATE Drug Patent Profile
✉ Email this page to a colleague
When do Meprobamate patents expire, and when can generic versions of Meprobamate launch?
Meprobamate is a drug marketed by Acella, Alembic Pharms Ltd, Barr, Chartwell Molecular, Elkins Sinn, Heather, Impax Labs, Invagen Pharms, Ivax Sub Teva Pharms, Lederle, Lee Km, Mallard, Mk Labs, Mylan, Nexgen Pharma Inc, Parke Davis, Perrigo, Pharmavite, Purepac Pharm, Pvt Form, Rising, Roxane, Sandoz, Scherer Labs, Solvay, Stanlabs Pharm, Sun Pharm Industries, Tablicaps, Taro, Usl Pharma, Valeant Pharm Intl, Vangard, Watson Labs, West Ward, Whiteworth Town Plsn, and Par Pharm. and is included in forty-seven NDAs.
The generic ingredient in MEPROBAMATE is aspirin; meprobamate. There are twenty-two drug master file entries for this compound. Additional details are available on the aspirin; meprobamate profile page.
Summary for MEPROBAMATE
| US Patents: | 0 |
| Applicants: | 36 |
| NDAs: | 47 |
| Finished Product Suppliers / Packagers: | 4 |
| Raw Ingredient (Bulk) Api Vendors: | 41 |
| Patent Applications: | 3,869 |
| Formulation / Manufacturing: | see details |
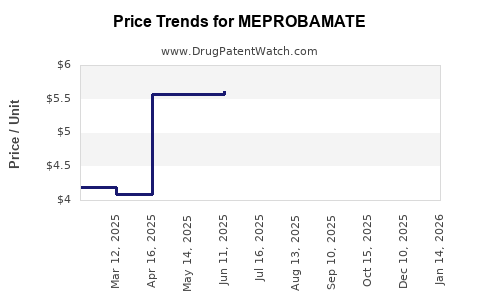
| Drug Prices: | Drug price information for MEPROBAMATE |
| DailyMed Link: | MEPROBAMATE at DailyMed |
Medical Subject Heading (MeSH) Categories for MEPROBAMATE
Anatomical Therapeutic Chemical (ATC) Classes for MEPROBAMATE
US Patents and Regulatory Information for MEPROBAMATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Stanlabs Pharm | MEPROBAMATE | meprobamate | TABLET;ORAL | 014474-004 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Purepac Pharm | MEPROBAMATE | meprobamate | TABLET;ORAL | 084804-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Valeant Pharm Intl | MEPROBAMATE | meprobamate | TABLET;ORAL | 015139-006 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


