MICONAZOLE NITRATE Drug Patent Profile
✉ Email this page to a colleague
When do Miconazole Nitrate patents expire, and when can generic versions of Miconazole Nitrate launch?
Miconazole Nitrate is a drug marketed by Perrigo R And D, Cosette, Perrigo, Taro, Teva, Teva Pharms, Actavis Pharma, P And L, and L Perrigo Co. and is included in twelve NDAs.
The generic ingredient in MICONAZOLE NITRATE is miconazole nitrate. There are twenty-eight drug master file entries for this compound. Forty-seven suppliers are listed for this compound. Additional details are available on the miconazole nitrate profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Miconazole Nitrate
A generic version of MICONAZOLE NITRATE was approved as miconazole nitrate by ACTAVIS PHARMA on November 19th, 1993.
Summary for MICONAZOLE NITRATE
| US Patents: | 0 |
| Applicants: | 9 |
| NDAs: | 12 |
| Finished Product Suppliers / Packagers: | 27 |
| Raw Ingredient (Bulk) Api Vendors: | 125 |
| Clinical Trials: | 7 |
| Patent Applications: | 3,137 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for MICONAZOLE NITRATE |
| DailyMed Link: | MICONAZOLE NITRATE at DailyMed |
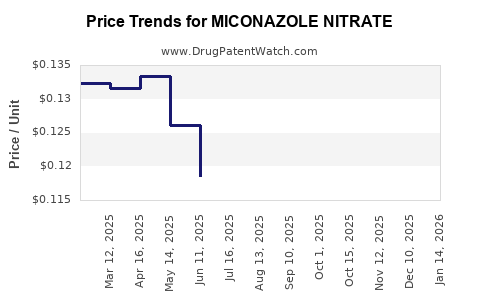
See drug prices for MICONAZOLE NITRATE
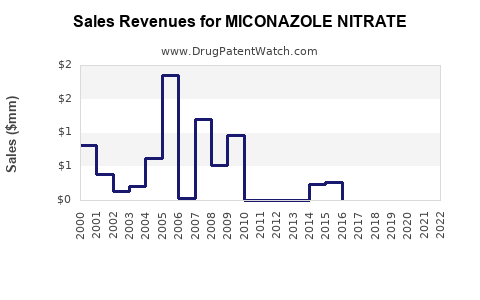
Recent Clinical Trials for MICONAZOLE NITRATE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Aesculape CRO Belgium BV | Phase 2 |
| Population Council | Phase 1 |
| Galeno Desenvolvimento de Pesquisas Clínicas | Phase 2 |
Pharmacology for MICONAZOLE NITRATE
| Drug Class | Azole Antifungal |
Anatomical Therapeutic Chemical (ATC) Classes for MICONAZOLE NITRATE
US Patents and Regulatory Information for MICONAZOLE NITRATE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Perrigo R And D | MICONAZOLE NITRATE | miconazole nitrate | CREAM, INSERT;TOPICAL, VAGINAL | 079114-001 | Jun 2, 2010 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva Pharms | MICONAZOLE NITRATE | miconazole nitrate | CREAM;VAGINAL | 074030-001 | Oct 30, 1992 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Perrigo R And D | MICONAZOLE NITRATE | miconazole nitrate | CREAM;VAGINAL | 091366-001 | Jan 15, 2010 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| L Perrigo Co | MICONAZOLE NITRATE COMBINATION PACK | miconazole nitrate | CREAM, SUPPOSITORY;TOPICAL, VAGINAL | 075329-001 | Apr 20, 1999 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Actavis Pharma | MICONAZOLE NITRATE | miconazole nitrate | SUPPOSITORY;VAGINAL | 073508-001 | Nov 19, 1993 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


