PANTOPRAZOLE SODIUM Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Pantoprazole Sodium, and when can generic versions of Pantoprazole Sodium launch?
Pantoprazole Sodium is a drug marketed by Ajanta Pharma Ltd, Annora Pharma, Dexcel, Sun Pharm, Acic Pharms, Aspiro, Be Pharms, Epic Pharma Llc, Eugia Pharma, Gland Pharma Ltd, Knack, Meitheal, Mylan Labs Ltd, Sandoz, Hikma, Actavis Totowa, Amneal Pharms, Aurobindo Pharma Ltd, Dr Reddys Labs Ltd, Granules, Hetero Labs Ltd V, Ingenus Pharms Llc, Jubilant Generics, L Perrigo Co, Lannett Co Inc, Macleods Pharms Ltd, Mankind Pharma, Mylan Pharms Inc, Orbion Pharms, Rubicon, Sun Pharm Inds Ltd, Teva, Torrent Pharms, Wockhardt Bio Ag, and Baxter Hlthcare Corp. and is included in thirty-seven NDAs.
The generic ingredient in PANTOPRAZOLE SODIUM is pantoprazole sodium. There are forty-eight drug master file entries for this compound. Sixty-nine suppliers are listed for this compound. Additional details are available on the pantoprazole sodium profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Pantoprazole Sodium
A generic version of PANTOPRAZOLE SODIUM was approved as pantoprazole sodium by DR REDDYS LABS LTD on January 19th, 2011.
Summary for PANTOPRAZOLE SODIUM
| US Patents: | 0 |
| Applicants: | 35 |
| NDAs: | 37 |
| Finished Product Suppliers / Packagers: | 67 |
| Raw Ingredient (Bulk) Api Vendors: | 67 |
| Clinical Trials: | 23 |
| Patent Applications: | 3,740 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for PANTOPRAZOLE SODIUM |
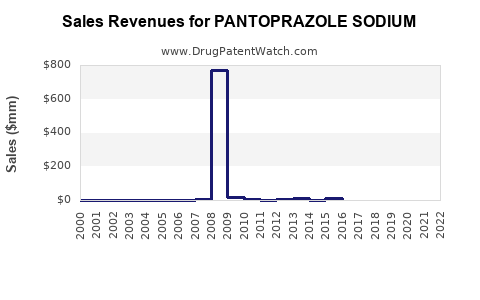
| Drug Sales Revenues: | Drug sales revenues for PANTOPRAZOLE SODIUM |
| What excipients (inactive ingredients) are in PANTOPRAZOLE SODIUM? | PANTOPRAZOLE SODIUM excipients list |
| DailyMed Link: | PANTOPRAZOLE SODIUM at DailyMed |
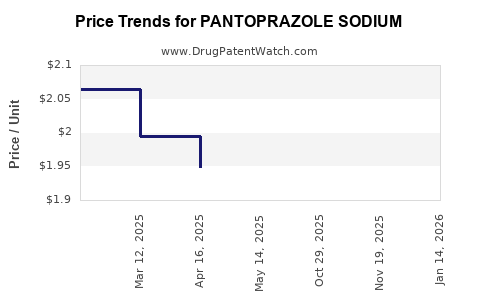
See drug prices for PANTOPRAZOLE SODIUM
Recent Clinical Trials for PANTOPRAZOLE SODIUM
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Alexandria University | Phase 3 |
| Mansoura University | Phase 1/Phase 2 |
| Fundação de Amparo à Pesquisa do Estado de São Paulo | Phase 4 |
Pharmacology for PANTOPRAZOLE SODIUM
| Drug Class | Proton Pump Inhibitor |
| Mechanism of Action | Proton Pump Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for PANTOPRAZOLE SODIUM
Paragraph IV (Patent) Challenges for PANTOPRAZOLE SODIUM
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| PROTONIX | for Delayed-release Oral Suspension | pantoprazole sodium | 40 mg | 022020 | 1 | 2019-09-13 |
| PROTONIX IV | For Injection | pantoprazole sodium | 40 mg/vial | 020988 | 1 | 2005-04-07 |
| PROTONIX | Delayed-release Tablets | pantoprazole sodium | 20 mg and 40 mg | 020987 | 2004-02-02 |
US Patents and Regulatory Information for PANTOPRAZOLE SODIUM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Orbion Pharms | PANTOPRAZOLE SODIUM | pantoprazole sodium | TABLET, DELAYED RELEASE;ORAL | 202052-002 | Dec 2, 2014 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| L Perrigo Co | PANTOPRAZOLE SODIUM | pantoprazole sodium | TABLET, DELAYED RELEASE;ORAL | 203024-001 | May 7, 2014 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva | PANTOPRAZOLE SODIUM | pantoprazole sodium | TABLET, DELAYED RELEASE;ORAL | 077056-002 | Aug 2, 2007 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


