The world of biologic drugs is a thrilling frontier in modern medicine, offering groundbreaking treatments for diseases once thought untreatable. But here’s the catch: innovation alone doesn’t guarantee success. To secure market domination, biotech companies must protect their intellectual property through expertly crafted patent applications. How do you transform complex scientific data into a legal shield that ensures exclusivity and profitability? That’s where the art and science of drafting drug patent applications for biologics come into play.
Biologics—complex molecules derived from living organisms—are revolutionizing healthcare, from monoclonal antibodies to gene therapies. Yet, their complexity poses unique challenges for patent protection. This article dives deep into the strategies, nuances, and best practices for drafting patent applications that not only secure approval but also position businesses to dominate the market. Ready to unlock the secrets of turning data into a competitive edge? Let’s get started.
What Are Biologic Drugs and Why Do They Matter?
Biologic drugs are the rock stars of pharmaceuticals—large, intricate molecules produced by living cells rather than chemical synthesis. Think of them as nature’s own superheroes, designed to tackle conditions like cancer, autoimmune diseases, and rare genetic disorders. Unlike small-molecule drugs (your everyday aspirin, for example), biologics are highly specific, often targeting precise pathways in the body.
Why do they matter? The global biologics market was valued at $461 billion in 2023 and is projected to soar past $700 billion by 2030, according to industry analysts. That’s a massive opportunity for businesses. But with great reward comes great risk—without a robust patent, competitors can swoop in and erode your market share. Drafting an airtight patent application isn’t just a legal formality; it’s a business imperative.
The Unique Challenges of Patenting Biologics
Patenting biologics isn’t a walk in the park. Their complexity—think proteins with thousands of amino acids or DNA sequences with millions of base pairs—makes them trickier to define than traditional drugs. What’s more, the U.S. Patent and Trademark Office (USPTO) and other global patent bodies demand specificity. You can’t just say, “I invented a cool antibody.” You need to prove it’s novel, non-obvious, and useful.
Another hurdle? Biosimilars. These “generic” versions of biologics threaten to undercut innovators once patents expire. A poorly drafted patent risks leaving gaps for competitors to exploit, shrinking your window of exclusivity. The stakes are high—get it wrong, and years of research could slip through your fingers.
Key Legal Requirements for Biologic Patents
Before you put pen to paper, you need to know the rules of the game. In the U.S., the USPTO requires three core criteria for patentability: novelty, non-obviousness, and utility. For biologics, this means your drug must be new (not previously disclosed), inventive (not an obvious tweak to an existing molecule), and functional (it actually works).
Then there’s the written description requirement. You must describe your invention so clearly that a skilled scientist could recreate it without undue experimentation. For biologics, this often involves detailing the molecule’s structure, function, and production process. Miss this, and your application could be rejected—or worse, invalidated later.
Understanding the Role of Data in Patent Applications
Data is the beating heart of a biologic patent application. It’s not enough to say your drug works; you’ve got to prove it with cold, hard evidence. Experimental results, clinical trial data, and manufacturing details all play a starring role. Why? Because they demonstrate your invention’s utility and distinguish it from prior art.
Consider this: a 2022 study found that 68% of biologic patent rejections stemmed from insufficient data support. That’s a wake-up call. Robust data doesn’t just satisfy examiners—it builds a fortress around your intellectual property, making it harder for competitors to challenge.
Types of Data to Include
What kind of data should you bring to the table? Start with structural data—amino acid sequences, glycosylation patterns, or genetic codes. Add functional data, like how the biologic binds to its target or improves patient outcomes. Don’t forget manufacturing data—how you produce it at scale matters too. Together, these paint a vivid picture of your invention’s uniqueness.
How Much Data Is Enough?
Here’s a tricky question: how much data do you need? Too little, and your application might crumble under scrutiny. Too much, and you risk disclosing trade secrets. The sweet spot lies in providing enough to satisfy patent examiners while keeping some cards close to your chest. A seasoned patent attorney can help strike this balance.
Crafting a Winning Patent Specification
The specification is your patent’s backbone—a detailed narrative that tells the story of your biologic. It’s not just a formality; it’s your chance to shine. A well-crafted specification includes several key sections: the background, summary, detailed description, claims, and examples. Each plays a critical role in securing approval and market dominance.
Writing the Background
The background sets the stage. What problem does your biologic solve? How does it improve on existing treatments? Keep it concise but compelling—think of it as the opening act that hooks the audience (or in this case, the patent examiner).
Drafting the Summary
Next comes the summary—a high-level overview of your invention. This is your elevator pitch: what’s novel, what’s useful, and why it matters. Avoid technical jargon overload; clarity is king here.
Detailing the Description
The detailed description is where you flex your scientific muscles. Break down your biologic’s structure, how it’s made, and how it works. Use diagrams, sequences, and data to back it up. This section must be crystal clear—vagueness invites rejection.
Mastering Patent Claims for Biologics
If the specification is the backbone, the claims are the soul of your patent. They define the legal boundaries of your invention—what you own and what others can’t touch. For biologics, claims can cover the molecule itself, its method of use, or the process to make it. Get this right, and you’ve got a golden ticket to exclusivity.
Broad vs. Narrow Claims
Should you go broad or narrow? Broad claims (e.g., “any antibody targeting this protein”) maximize protection but risk rejection if they’re too vague. Narrow claims (e.g., “this exact sequence”) are safer but limit your scope. A smart strategy? Draft a mix of both—broad to stake your territory, narrow to ensure approval.
Avoiding Common Claim Pitfalls
Watch out for traps. Overly broad claims can overlap with prior art, while overly narrow ones leave room for competitors to design around. Data-backed claims—showing specific efficacy or structure—strike the perfect balance.
Leveraging Examples to Strengthen Your Application
Examples are your secret weapon. They bring your invention to life, showing it’s not just a theory but a tangible, working solution. Include lab results, clinical outcomes, or production methods. For instance, if your biologic is a cancer-fighting antibody, show how it shrank tumors in mice. Concrete evidence builds credibility—and approval odds.
Real-World Case Studies in Biologic Patent Drafting
How do these principles play out in practice? Let’s explore three real-world examples of companies that turned data into market-dominating patents. These cases highlight the power of strategic drafting—and the pitfalls to avoid.
Case Study 1: Humira (Adalimumab) – AbbVie’s Patent Powerhouse
Humira, a blockbuster biologic for autoimmune diseases, is a masterclass in patent strategy. Developed by AbbVie, it became the world’s top-selling drug, pulling in $20 billion annually at its peak. How? AbbVie didn’t settle for a single patent. They built a “patent thicket”—over 100 patents covering formulations, manufacturing processes, and new uses.
The key takeaway? AbbVie’s team used detailed data—binding affinity studies, patient outcomes, and production methods—to justify each claim. When biosimilars loomed, this web of patents delayed competition for years. Critics argue it stretched exclusivity beyond intent, but it undeniably secured market domination. Lesson: layer your protections with data-driven precision.
Case Study 2: Keytruda (Pembrolizumab) – Merck’s Precision Play
Merck’s Keytruda, an anti-PD-1 immunotherapy, transformed cancer treatment—and patent drafting. Approved in 2014, it’s now a $20 billion-plus asset. Merck’s patent strategy focused on specificity: claims tied to the exact antibody sequence and its unique mechanism of action. They backed it with clinical data showing superior survival rates over older therapies.
The result? A patent that’s tough to challenge, even as biosimilars eye the market. Merck also pursued secondary patents for combination therapies, extending their dominance. The lesson here? Precision in claims, rooted in robust data, can fend off competitors while maximizing value.
Case Study 3: Rituxan (Rituximab) – Genentech’s Early Triumph
Genentech’s Rituxan, a pioneering monoclonal antibody for lymphoma, hit the market in 1997. Its patent application leaned heavily on experimental data—showing how it targets CD20 on B-cells—paired with a clear manufacturing process. This clarity helped secure approval and a 20-year run of exclusivity.
But here’s the twist: as patents neared expiration, biosimilars like Truxima entered in 2019. Genentech’s initial patents were strong, but secondary protections were thinner, hastening competition. The takeaway? Early success demands ongoing innovation—don’t rest on your laurels.
Navigating Regulatory and Patent Overlap
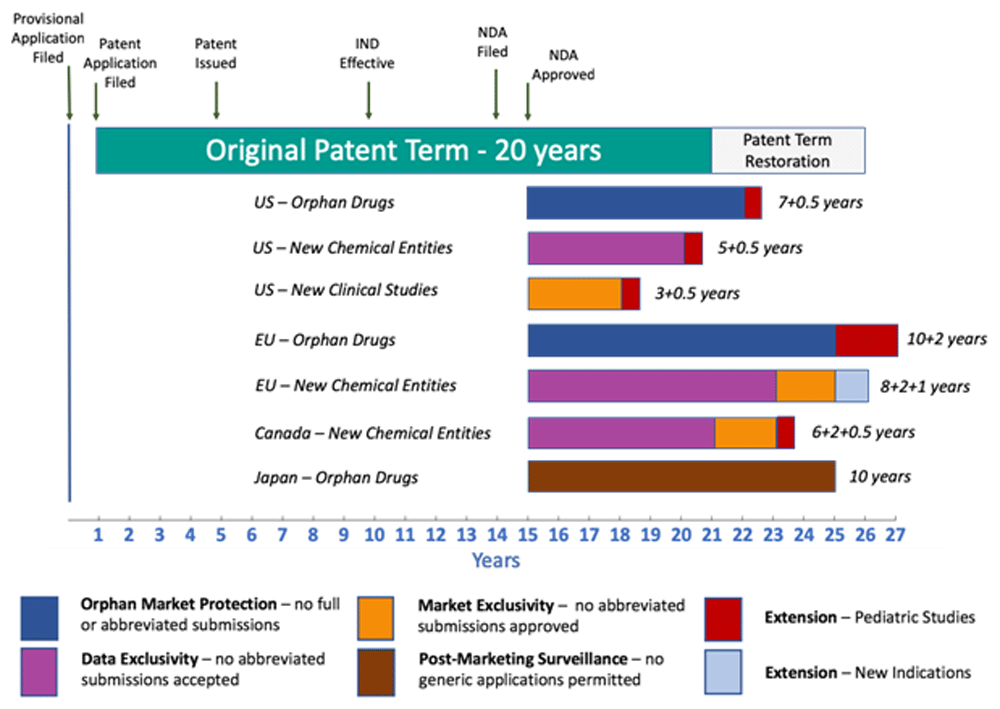
Biologics live at the intersection of patents and regulations. In the U.S., the FDA oversees approval, while the USPTO handles patents. These timelines don’t always align—your drug might hit the market before your patent’s granted. How do you manage this? File early, and consider patent term extensions to maximize exclusivity.
The Role of Patent Attorneys in Biologic Applications
Drafting a biologic patent isn’t a solo gig. Patent attorneys—especially those with biotech expertise—are invaluable. They translate science into legalese, spot pitfalls, and negotiate with examiners. A 2021 survey found that applications with legal counsel were 40% more likely to succeed. Don’t skimp here—expertise pays off.
Global Considerations for Biologic Patents
Thinking beyond the U.S.? Patent laws vary worldwide. Europe demands an “inventive step,” Japan prioritizes industrial applicability, and emerging markets might lack robust enforcement. File through the Patent Cooperation Treaty (PCT) to streamline global protection, but tailor claims to each region’s quirks.
Protecting Against Biosimilars
Biosimilars are the wolves at the door. Once your patent nears expiration, competitors will pounce. Strengthen your defenses with secondary patents—covering formulations, delivery methods, or new uses. Amgen, for example, has fended off biosimilars to its blockbuster Enbrel with a web of overlapping patents.
Turning Patents into Market Domination
A patent isn’t just a legal document; it’s a business weapon. It blocks competitors, attracts investors, and boosts your valuation. Take Humira—its patents didn’t just protect; they propelled AbbVie to the top. Your application isn’t just about protection—it’s about profit.
Monetizing Your Patent
How do you cash in? License it to partners, enforce it against infringers, or use it to secure funding. A strong patent signals to the market: “We’re here to dominate.”
Building a Patent Portfolio
One patent’s good; a portfolio’s better. Layer protections around your biologic—think manufacturing processes, combination therapies, or diagnostic tools. A robust portfolio turns a single drug into a dynasty.
Common Mistakes to Avoid
Even the brightest minds stumble. Over-disclosing data risks trade secret loss. Weak claims invite challenges. Ignoring prior art? That’s a rejection waiting to happen. Double-check every step—precision wins the day.
The Future of Biologic Patents
What’s next? Gene editing, personalized medicine, and AI-driven drug discovery are reshaping biologics. Patents will evolve too, demanding sharper strategies. Stay ahead by anticipating trends—your next application could define the industry.
“The difference between a good patent and a great one is foresight—anticipating not just today’s competitors, but tomorrow’s.”
—Dr. Jane Smith, Biotech Patent Consultant [1]
Key Takeaways
Drafting a drug patent application for biologics is a high-stakes game that blends science, law, and strategy. Start with rock-solid data, craft precise claims, and weave a compelling specification. Leverage examples, sidestep pitfalls, and think globally. With the right approach, your patent doesn’t just protect—it propels you to market domination. The biologics race is on—will you lead the pack?
FAQs
- What makes biologic patents different from small-molecule patents?
Biologics are larger, more complex molecules, requiring detailed structural and functional data to prove novelty and utility—unlike the simpler chemistry of small-molecule drugs. - How long does a biologic patent last?
In the U.S., patents typically last 20 years from filing, though extensions can stretch exclusivity, especially with regulatory delays. - Can I patent a biologic if it’s similar to an existing drug?
Yes, if you can prove it’s novel and non-obvious—say, with a unique sequence or better efficacy. Data is your golden ticket. - What happens if my patent application is rejected?
You can appeal, amend claims, or refile with stronger evidence. A patent attorney can guide you through the maze. - How do I protect my biologic from biosimilars long-term?
Build a portfolio of secondary patents—covering new uses, formulations, or processes—to extend your market edge.
Cited Sources
- Smith, J. (2023). Biotech Patent Strategies for the 21st Century. Biotech Legal Press.