Enhancing Your Product Portfolio with CDMOs: A Strategic Partnership for Pharmaceutical Success

The pharmaceutical industry is constantly evolving, driven by advancements in technology, innovations in product development, and the need for efficient manufacturing processes. In this dynamic landscape, Contract Development and Manufacturing Organizations (CDMOs) have emerged as crucial partners for pharmaceutical companies, offering a range of services that support the development and production of pharmaceutical and biopharmaceutical products. By leveraging the expertise and resources of CDMOs, pharmaceutical companies can focus on their core competencies while outsourcing various stages of drug development and manufacturing.
Expertise and Flexibility
CDMOs provide expertise in areas such as drug discovery and development, clinical trials, process development and scale-up, and commercial-scale manufacturing. They offer a flexible approach to drug development and manufacturing, allowing them to rapidly scale up or down to meet the changing needs of their clients. This adaptability is particularly valuable for small and emerging pharmaceutical companies that may not have the resources or expertise to develop and manufacture their own products.
Cost Management and Efficiency
Partnering with a CDMO can help pharmaceutical companies manage costs by leveraging their economies of scale and optimizing processes. CDMOs can reduce waste, streamline development and manufacturing timelines, and ensure regulatory compliance. This not only saves costs but also accelerates time to market, allowing companies to bring new products to market more efficiently.
Scalability and Supply Chain Management
CDMOs operate on a large scale, managing inventory and sourcing materials from a global network of suppliers. They have the capacity to handle projects of varying sizes, from small clinical trials to large-scale manufacturing of generic drugs. This scalability is essential for pharmaceutical companies, as it allows them to adapt to changing market demands and production volumes.
Regulatory Compliance and Quality Control
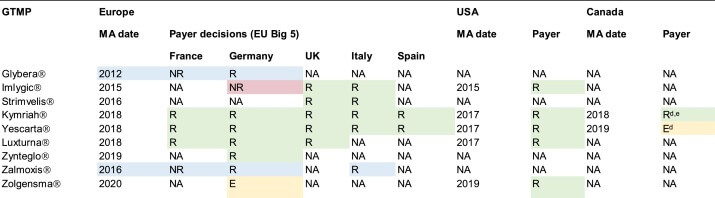
CDMOs are experts in regulatory compliance and quality control, ensuring that products meet the highest standards of quality, safety, and efficacy. They have the necessary expertise and equipment to handle complex manufacturing processes, including novel modalities such as cell therapies, gene therapies, and mRNA therapies.
Strategic Partnerships
CDMOs are not just service providers; they are strategic partners that can help pharmaceutical companies increase their competitiveness. By outsourcing to CDMOs, companies can reduce their capital expenses, vendor management costs, and inventory management costs. CDMOs also help control supply risk by maintaining robust business continuity plans and flexible supply chains.
Case Studies and Industry Insights
Industry experts highlight the importance of CDMOs in the pharmaceutical industry. For instance, Vincent Wong, Chief Quality Officer at TOMZ Corporation, emphasizes the value of CDMOs in providing design for manufacturing (DfM) services, which are critical for medtech and meddev companies. Similarly, a report by EY-Parthenon notes that CDMOs are becoming emerging technology leaders, driving innovation in the pharmaceutical industry through their expertise and capabilities.
Conclusion
In conclusion, CDMOs play a vital role in enhancing the product portfolio of pharmaceutical companies. By offering expertise, flexibility, and efficiency, CDMOs help companies reduce costs, accelerate time to market, and ensure regulatory compliance. As the pharmaceutical industry continues to evolve, the importance of CDMOs will only grow, providing a strategic partnership for pharmaceutical companies to succeed.
“CDMOs are not just service providers; they are strategic partners that can help pharmaceutical companies increase their competitiveness.” – Borac DMO
References
- Mantell Associates. (2023, February). The Increasing Importance of CDMOs in the Pharmaceutical Industry.
- EY. (n.d.). How CDMOs are leading innovation for pharmaceutical partners.
- MedPak. (n.d.). What is a CDMO & How Do They Help Pharma Companies?
- Borac DMO. (n.d.). How a CDMO Can Increase the Competitiveness of Your Product.
- Propel Software. (2022, November). How CDMOs are evolving to enrich medical device manufacturing.