[](//www.DrugPatentWatch.com/p/preview/generic-api/eltrombopag?utm_medium=dpw_wp_blog&utm_campaign=dpw_wp_blog&utm_source=dpw_wp_blog)
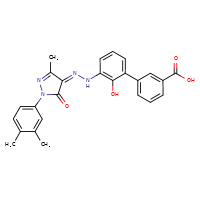
Eltrombopag is the generic ingredient in two branded drugs marketed by Novartis and is included in two NDAs. There are eight patents protecting this compound.
There are four tentative approvals for this compound.
For more information on how DrugPatentWatch can help with your pharmaceutical business intelligence needs, contact admin@DrugPatentWatch.com or visit www.DrugPatentWatch.com
