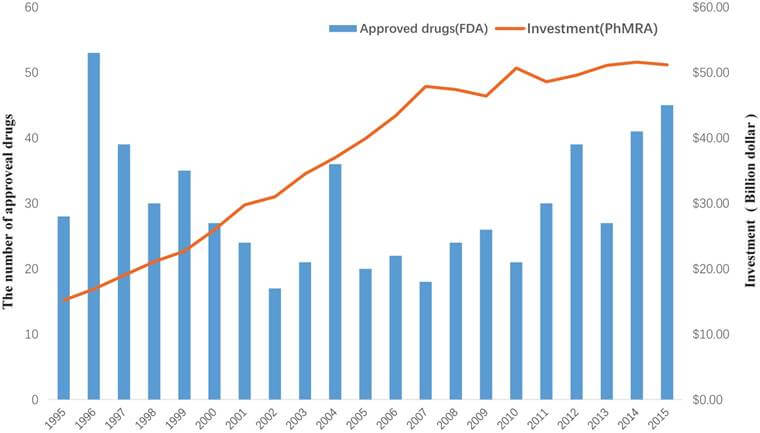
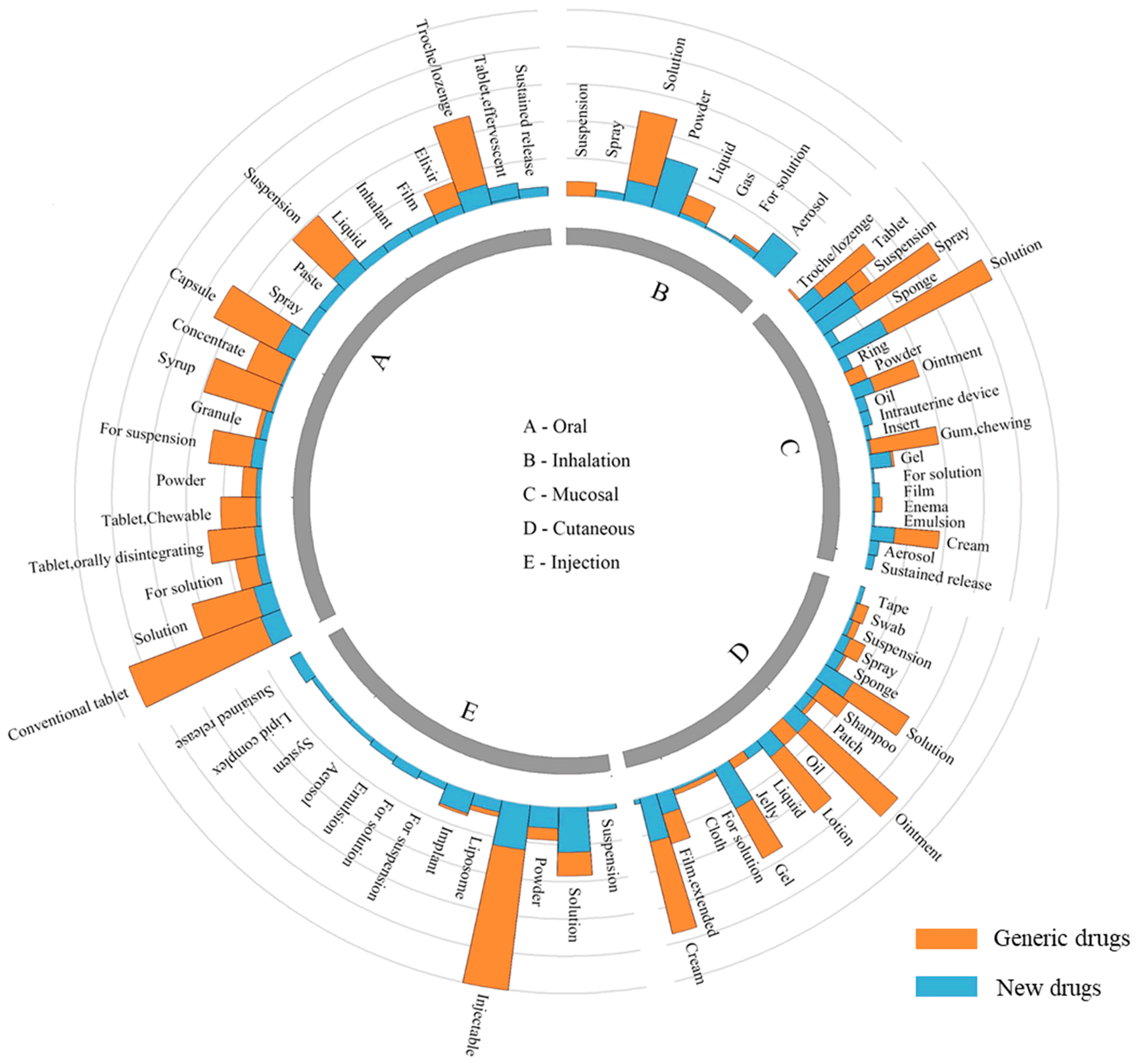
The pharmaceutical industry has increasingly turned to drug combinations as a means to develop more effective treatments for various diseases. These combinations often involve the co-administration of two or more active ingredients, which can lead to enhanced therapeutic effects, reduced side effects, or improved patient compliance. However, patenting these drug combinations can be a complex process, requiring careful consideration of various legal and scientific factors.
The Concept of Combination Inventions
A combination invention involves the linking of two or more active ingredients by a functional interaction, resulting in a combined technical effect that is different from the sum of the effects produced by each ingredient individually. This combined technical effect can manifest in various ways, such as a synergistic effect, reduced side effects, or improved bioavailability. To satisfy the criterion of inventive step, this technical effect must not be suggested by the prior art.
Patenting Strategies
Several strategies can be employed to extend patent protection for drug combinations:
New Formulations: Patenting new formulations of a known compound can provide additional protection. This includes different dosage forms, regimens, or co-formulations with other pharmaceuticals.
Combination Products: Patenting combination products, such as dual-barrel syringes, or co-administration of different products simultaneously, can also provide extended protection.
Secondary Patents: Filing secondary patents for technological advances related to the drug, such as new formulations or combination therapies, can significantly extend the period of patent protection.
Challenges in Patenting Drug Combinations
Despite these strategies, patenting drug combinations can be challenging due to several factors:
Obviousness: The co-administration of two or more medicines to treat a disease is often a routine medical practice. This can make it difficult to demonstrate that a combination is not obvious to a skilled person in the relevant technical field.
Prior Art: The European Patent Office (EPO) assesses the obviousness of an invention based on prior art documents. If an applicant cannot demonstrate that a combination is particularly efficacious, a patent may not be granted.
Evergreening: Pharmaceutical companies may employ strategies aimed at extending the period of protection through minor modifications to a drug’s dosage, formulation, or delivery system. This practice, known as evergreening, can lead to financial and patient care costs on society.
Expert Insights
“Developing successful drug combinations requires more than just pharmaceutical knowledge. Build a team that includes members with expertise in various fields, including law, to ensure that all aspects of the combination are properly protected.” – PatentPC
“Combination inventions must demonstrate a functional interaction between the active ingredients, leading to a combined technical effect that is different from the sum of the effects produced by each ingredient individually.” – Lexology
Conclusion
Patenting drug combinations is a complex process that requires careful consideration of various legal and scientific factors. By employing strategies such as new formulations, combination products, and secondary patents, pharmaceutical companies can extend patent protection for their drug combinations. However, challenges such as obviousness and evergreening must be addressed to ensure that these patents are robust and provide meaningful protection.
References
- PatentPC. (2023, September 7). How to Patent Drug Combinations and Cocktails. Retrieved from https://patentpc.com/blog/how-to-patent-drug-combination-and-cocktails
- Lexology. (2021, December 30). Protecting a drug combining two known active ingredients by patent. Retrieved from https://www.lexology.com/library/detail.aspx?g=26ccfaeb-2c33-41bc-a361-ea042533b7e6
- NCBI. (n.d.). Patent protection strategies. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3146086/
- Penningtonslaw. (2022, September 23). Patents to biological medicines in combination: is two really better than one? Retrieved from https://www.penningtonslaw.com/news-publications/latest-news/2022/patents-to-biological-medicines-in-combination-is-two-really-better-than-one
- Nature. (2021, December 8). Combination therapy patents: a new front in evergreening. Retrieved from https://www.nature.com/articles/s41587-021-01137-6