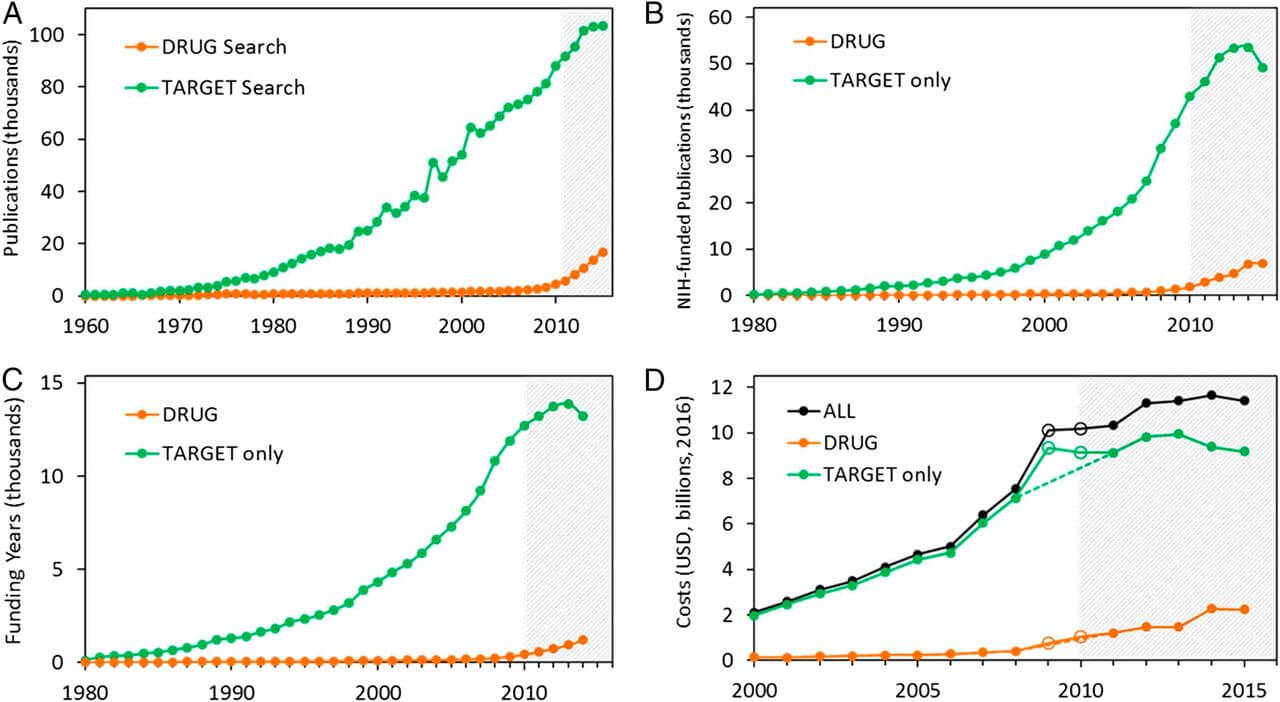
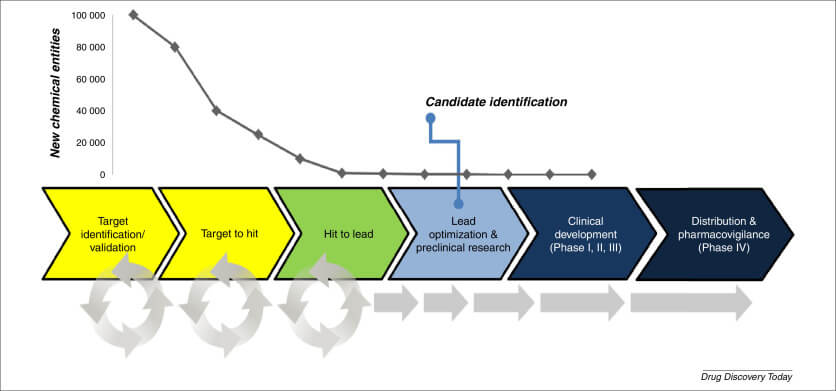
Pharmacovigilance, the science and activities related to the detection, assessment, understanding, and prevention of adverse effects or any other drug-related problem, plays a crucial role in ensuring the safety and efficacy of generic drugs. Generic drugs are expected to be cost-effective and have the same therapeutic effect as their brand-name counterparts. However, the development and approval process for generics involves a two-tiered regulatory framework of pharmaceutical equivalence and bioequivalence, which can sometimes lead to differences in quality and user interfaces that may impact safety and efficacy.
Ensuring Safety Before Approval
The FDA’s Office of Surveillance and Epidemiology (OSCE) plays a critical role in ensuring the safety of generic drugs even before they are approved. During the application review process, generic drug applicants must demonstrate that their product is the same as the brand-name drug. OSCE evaluates safety submissions to assess whether there are any unexpected safety findings that could raise concerns about the proposed generic drug. This includes the timely submission of serious adverse event reports that occur during investigational generic drug bioequivalence studies.
Post-Marketing Surveillance
After a generic drug is approved, OSCE conducts pharmacovigilance screening to detect any early signals of potential safety concerns. This involves monitoring and evaluating postmarketing reports, including those submitted to FDA’s MedWatch program, which helps track potential new safety or quality issues. OSCE also evaluates potential safety concerns by using published scientific literature reviews, direct contact from patients or healthcare professionals to FDA, manufacturer reports, internal FDA databases, and market share data.
Addressing Safety Concerns
If OSCE identifies a safety concern before approval, it works with the applicant to gather additional information or make changes to the generic drug to ensure it has the same safety profile as the brand-name drug. If a concern is identified after approval, OSCE reviews all relevant data to take appropriate action to safeguard public health. This may lead to changes in how a generic drug is used or manufactured, such as drug recalls or reformulations to address issues.
Challenges in Pharmacovigilance
Pharmacovigilance for generic drugs faces several challenges. For instance, the high number of perceived therapeutic failure reports combined with high rates of generic drug dispensing, general underreporting of postmarketing adverse events, and incomplete information provided in adverse event reports can make it difficult to determine whether reports of perceived therapeutic failure indicate a real difference in effectiveness between an RLD and one or more of its generics.
Importance of Collaboration
Pharmacovigilance is a collaborative effort involving patients, healthcare professionals, regulatory agencies, and pharmaceutical companies. Patients and healthcare professionals play a key role in reporting adverse reactions or quality problems to FDA’s MedWatch program, which helps track potential new safety or quality issues. Regulatory agencies like the FDA work with pharmaceutical companies to ensure that they comply with safety regulations and reporting requirements.
Conclusion
Pharmacovigilance is essential for ensuring the safety and efficacy of generic drugs. The FDA’s Office of Surveillance and Epidemiology plays a critical role in evaluating safety submissions and conducting postmarketing surveillance to detect potential safety concerns. Collaboration between patients, healthcare professionals, regulatory agencies, and pharmaceutical companies is crucial for maintaining the quality, efficacy, and safety of generic drugs in the U.S. market.
References
- FDA. (2023). Ensuring the Safety of FDA-Approved Generic Drugs. Retrieved from https://www.fda.gov/drugs/cder-conversations/ensuring-safety-fda-approved-generic-drugs
- FDA. (2018). Overview of the Generic Drug Program and Surveillance. Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6027587/
- Faculty of Medicine, Gadja Mada University. (n.d.). Importance of Pharmacovigilance for Pharmaceutical Industry. Retrieved from https://www.pmda.go.jp/files/000216986.pdf
- Oza, B., Radhakrishna, S., Pipalava, P., & Jose, V. (2020). Pharmacovigilance of biosimilars – Why is it different from generics and innovator biologics? Retrieved from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6813686/
- Forsyth, L., Kim, E., & Catterson, D. (2020). The Importance of Generic Drug Pharmacovigilance. Retrieved from https://www.youtube.com/watch?v=F2rJ-XnKkBs