Digital twins are revolutionizing the pharmaceutical and biopharmaceutical industries by integrating physical plants, data collection, data analysis, and system control. This integration enables real-time monitoring and optimization of manufacturing processes, leading to improved product quality, reduced waste, and enhanced regulatory compliance.
Digital Twins in Generic Drug Manufacturing
Generic drug manufacturing is a crucial aspect of the pharmaceutical industry, where the focus is on producing high-quality, cost-effective versions of existing medications. Digital twins can significantly contribute to this process by providing predictive analytics and real-time monitoring capabilities.
Predictive Analytics
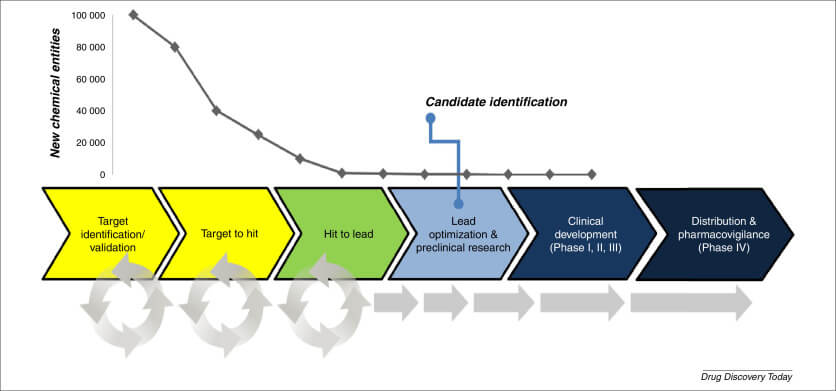
Digital twins can simulate various scenarios and predict the performance of different manufacturing processes. This predictive capability helps generic drug manufacturers optimize their processes, reducing the time and cost associated with physical experiments. For instance, digital twins can predict product quality, productivity, and process attributes, enabling manufacturers to make informed decisions about their production routes.
Real-Time Monitoring
Real-time monitoring is a critical aspect of digital twins in generic drug manufacturing. By integrating IoT sensors and advanced data analytics, digital twins can provide real-time insights into the manufacturing process. This allows production managers to identify and address any issues promptly, ensuring that the final product meets the required quality standards.
Regulatory Compliance
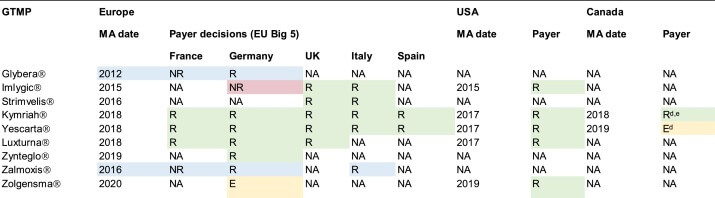
Digital twins can also enhance regulatory compliance in generic drug manufacturing. By providing detailed records of the manufacturing process, digital twins can help manufacturers demonstrate compliance with regulatory requirements. This is particularly important in the pharmaceutical industry, where regulatory compliance is critical for ensuring patient safety and maintaining market credibility.
Case Studies
Several companies are already leveraging digital twins in their generic drug manufacturing processes. For example, Atos has developed a digital twin solution for vaccine development, which has shown significant improvements in reducing the time to market for new drugs.
Challenges and Opportunities
While digital twins offer numerous benefits in generic drug manufacturing, there are also challenges to be addressed. For instance, the integration of digital twins into existing manufacturing processes can be complex and require significant investments in infrastructure and training. Additionally, there are regulatory hurdles to overcome, as digital twins must comply with existing regulatory frameworks.
Conclusion
Digital twins are transforming the pharmaceutical and biopharmaceutical industries by providing real-time monitoring, predictive analytics, and regulatory compliance. In generic drug manufacturing, digital twins can significantly improve product quality, reduce waste, and enhance regulatory compliance. As the industry continues to evolve, the role of digital twins will become even more crucial in ensuring the efficient and effective production of high-quality generic drugs.
“Digital twins can significantly improve product quality, reduce waste, and enhance regulatory compliance in generic drug manufacturing.”
References
- Chen, Y., Yang, O., Sampat, C., Bhalode, P., Ramachandran, R., & Ierapetritou, M. (2020). Digital Twins in Pharmaceutical and Biopharmaceutical Manufacturing: A Literature Review. Processes, 8(9), 1088. https://doi.org/10.3390/pr8091088
- Kritzinger, W., Karner, M., Traar, G., Henjes, J., & Sihn, W. (2018). Digital Twin in manufacturing: A categorical literature review and classification. IFAC-PapersOnLine, 51(1), 1016–1022. [Google Scholar] [CrossRef]
- Oztemel, E., & Gursev, S. (2018). Literature review of Industry 4.0 and related technologies. Journal of Intelligent Manufacturing, 31(1), 127–182. [Google Scholar] [CrossRef]
- Tao, F., Cheng, J., Qi, Q., Zhang, M., Zhang, H., & Sui, F. (2018). Digital twin-driven product design, manufacturing and service with big data. International Journal of Advanced Manufacturing Technology, 94(1), 3563–3576. [Google Scholar] [CrossRef]
- Venkatasubramanian, V. (2019). The promise of artificial intelligence in chemical engineering: Is it here, finally? AIChE Journal, 65(1), 466–478. [Google Scholar] [CrossRef]
- Bao, J., Guo, D., Li, J., & Zhang, J. (2018). The modelling and operations for the digital twin in the context of manufacturing. Enterprise Information Systems, 13(3), 534–556. [Google Scholar] [CrossRef]
- Tao, F., Qi, Q., Wang, L., & Nee, A.Y.C. (2019). Digital Twins and Cyber–Physical Systems toward Smart Manufacturing and Industry 4.0: Correlation and Comparison. Engineering, 5(1), 653–661. [Google Scholar] [CrossRef]