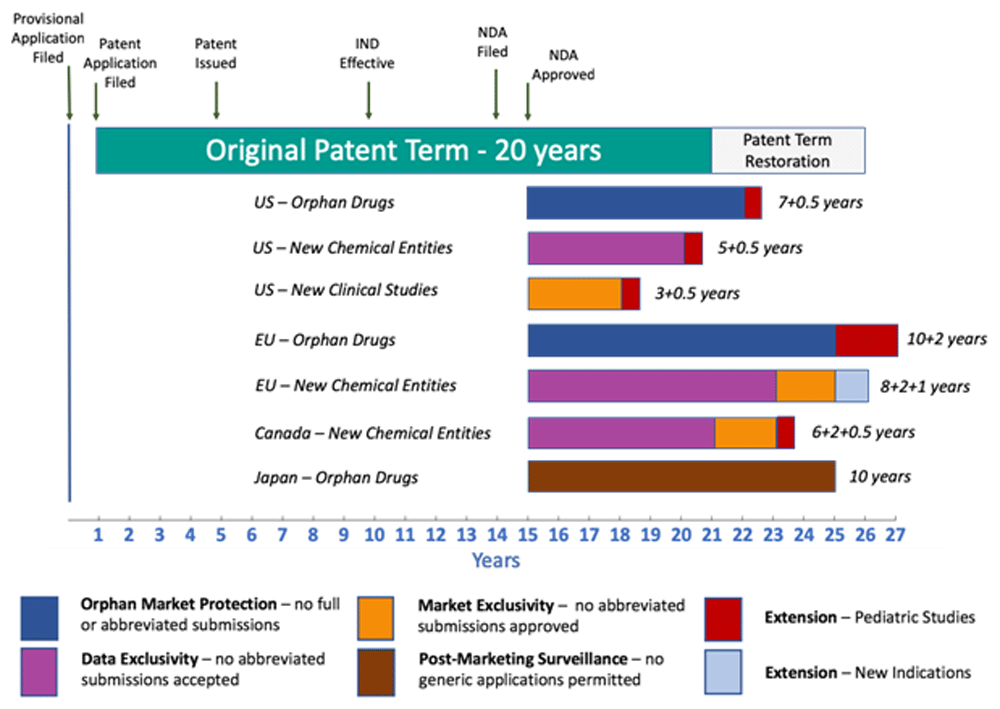
The pharmaceutical industry relies heavily on quality assurance (QA) to ensure that generic drugs meet the same standards of quality, safety, and efficacy as their brand-name counterparts. QA plays a crucial role in guaranteeing that generic drugs are manufactured to the highest standards, thereby maintaining patient trust and confidence in these cost-effective alternatives.
Ensuring Pharmaceutical Equivalence and Bioequivalence
Generic drugs must demonstrate pharmaceutical equivalence and bioequivalence to the brand-name drug they are intended to replace. Pharmaceutical equivalence ensures that the active ingredient, strength, dosage form, and route of administration are the same in both drugs. Bioequivalence, on the other hand, ensures that there is no significant difference in the rate and extent of absorption between the generic and brand-name drugs.
FDA Oversight and Approval Process
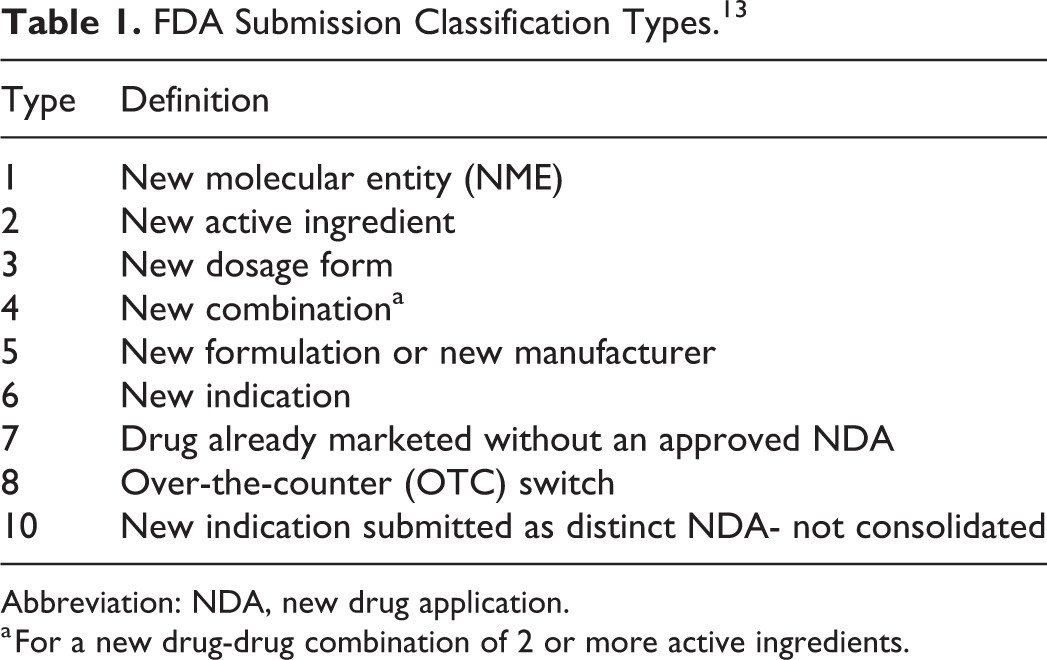
The U.S. Food and Drug Administration (FDA) is responsible for ensuring that generic drugs meet these stringent standards. The FDA’s review process involves intensive assessments prior to approval, including rigorous testing and inspections of manufacturing facilities. Generic drugs manufactured outside the U.S. must also meet the same approval standards as those made domestically.
Quality Control Issues and Challenges
Despite the FDA’s oversight, quality control issues have been reported in the generic drug market. For instance, recalls of generic drugs have been necessary due to contamination with carcinogens, highlighting the need for more robust quality control measures. The globalization of the supply chain has also made it more challenging for the FDA to maintain quality oversight, as many generic drugs are now manufactured overseas.
The Need for a Rating System and Independent Testing
Experts argue that a rating system for generic drugs, similar to those used for consumer products, could help consumers make informed decisions about the quality of the medications they use. Independent testing of generic drugs at various stages of the supply chain could also provide a more comprehensive picture of their quality.
Conclusion
In conclusion, quality assurance is a critical component of the generic drug industry, ensuring that these cost-effective medications meet the same standards of quality, safety, and efficacy as their brand-name counterparts. While the FDA plays a vital role in overseeing the approval process, quality control issues and challenges persist. Implementing a rating system and independent testing could help address these issues and provide consumers with greater confidence in the quality of generic drugs.
References
Quanta. (2024, March 27). Quality control vs quality assurance in pharmaceuticals. Retrieved from https://www.quanta-cs.com/blogs/2024-3/quality-control-vs-quality-assurance-pharma
American Academy of Family Physicians. (2018, June 1). How the FDA ensures high-quality generic drugs. Retrieved from https://www.aafp.org/pubs/afp/issues/2018/0601/p696.html
Circulation. (2022, April 18). Price and quality in the generic pharmaceutical market. Retrieved from https://www.ahajournals.org/doi/10.1161/CIRCULATIONAHA.121.057727
WFAE. (2021, October 5). Generic drugs are cheaper in the U.S., but quality control can be a problem, professor says. Retrieved from https://www.wfae.org/health/2021-10-05/generic-drugs-are-cheaper-in-the-u-s-but-quality-control-can-be-a-problem-professor-says
U.S. Food and Drug Administration. (2019, May 13). Safety, efficacy, and quality remain top priorities as we continue our work to expand access to cost-saving generic drugs. Retrieved from https://www.fda.gov/news-events/fda-voices/safety-efficacy-and-quality-remain-top-priorities-we-continue-our-work-expand-access-cost-saving