Doxycycline - Generic Drug Details
✉ Email this page to a colleague
What are the generic drug sources for doxycycline and what is the scope of patent protection?
Doxycycline
is the generic ingredient in eighteen branded drugs marketed by Alembic, Apotex, Cosette, Dr Reddys, Dr Reddys Labs Sa, Impax Labs Inc, Lupin, Lupin Ltd, Rising, Sandoz Inc, Strides Pharma, Sun Pharm Inds Ltd, Watson Labs, Zydus Pharms, Chartwell Rx, Galderma Labs Lp, Rachelle, Chartwell, Pfizer, Heritage, Lannett Co Inc, Somerset Theraps Llc, Sun Pharm Industries, Pliva, Mayne Pharma, Warner Chilcott, Bausch, Teva, Actavis Labs Fl Inc, Ajanta Pharma Ltd, Amneal Pharms, Changzhou Pharm, Halsey, Heather, Hikma Intl Pharms, Interpharm, Mutual Pharm, Nesher Pharms, Nostrum Labs Inc, Par Pharm, Pvt Form, Ranbaxy, Superpharm, Zhejiang Yongtai, Zydus Lifesciences, Collagenex, Fresenius Kabi Usa, Hikma, Mylan Labs Ltd, Endo Operations, Gland Pharma Ltd, Slate Run Pharma, West-ward Pharms Int, Den-mat, Actavis Elizabeth, Aurobindo Pharma Usa, Prinston Inc, Acella, Amneal, Amneal Pharms Co, Avet Lifesciences, Caribe Holdings, Chartwell Molecular, Epic Pharma Llc, Heritage Pharma, Larken Labs, Mylan, Novel Labs Inc, Oryza, Praxgen, Torrent, and Chartwell Pharma, and is included in one hundred and thirty-three NDAs. There are ten patents protecting this compound and one Paragraph IV challenge. Additional information is available in the individual branded drug profile pages.Doxycycline has eighteen patent family members in eight countries.
There are twenty-eight drug master file entries for doxycycline. Forty-three suppliers are listed for this compound. There are five tentative approvals for this compound.
Summary for doxycycline
| International Patents: | 18 |
| US Patents: | 10 |
| Tradenames: | 18 |
| Applicants: | 72 |
| NDAs: | 133 |
| Drug Master File Entries: | 28 |
| Finished Product Suppliers / Packagers: | 43 |
| Raw Ingredient (Bulk) Api Vendors: | 64 |
| Clinical Trials: | 364 |
| Patent Applications: | 6,809 |
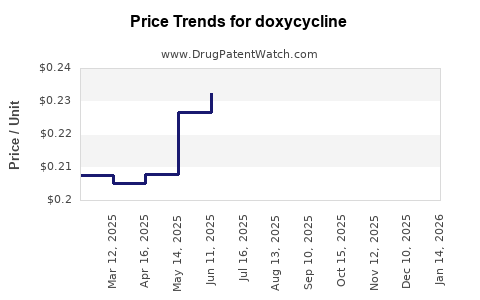
| Drug Prices: | Drug price trends for doxycycline |
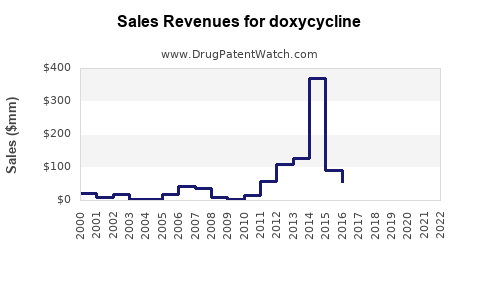
| Drug Sales Revenues: | Drug sales revenues for doxycycline |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for doxycycline |
| What excipients (inactive ingredients) are in doxycycline? | doxycycline excipients list |
| DailyMed Link: | doxycycline at DailyMed |
Recent Clinical Trials for doxycycline
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Chinese University of Hong Kong | Phase 4 |
| Lao-Oxford-Mahosot Hospital Wellcome Trust Research Unit | Phase 2/Phase 3 |
| Mahidol Oxford Tropical Medicine Research Unit | Phase 2/Phase 3 |
Generic filers with tentative approvals for DOXYCYCLINE
The 'tentative' approval signifies that the product meets all FDA standards for marketing, and, but for the patents / regulatory protections, it would approved.
Pharmacology for doxycycline
| Drug Class | Tetracycline-class Drug |
Medical Subject Heading (MeSH) Categories for doxycycline
Anatomical Therapeutic Chemical (ATC) Classes for doxycycline
Paragraph IV (Patent) Challenges for DOXYCYCLINE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| ORACEA | Delayed-release Capsules | doxycycline | 40 mg | 050805 | 1 | 2008-12-12 |
US Patents and Regulatory Information for doxycycline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mayne Pharma | DORYX MPC | doxycycline hyclate | TABLET, DELAYED RELEASE;ORAL | 050795-008 | May 20, 2016 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Interpharm | DOXYCYCLINE HYCLATE | doxycycline hyclate | CAPSULE;ORAL | 062763-002 | Sep 2, 1988 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Ajanta Pharma Ltd | DOXYCYCLINE HYCLATE | doxycycline hyclate | TABLET;ORAL | 211584-002 | Jun 1, 2020 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Heritage | DOXYCYCLINE HYCLATE | doxycycline hyclate | INJECTABLE;INJECTION | 217854-001 | Jul 23, 2024 | AP | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Heritage | DOXYCYCLINE | doxycycline | TABLET;ORAL | 091605-003 | Dec 20, 2011 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Strides Pharma | DOXYCYCLINE | doxycycline | CAPSULE;ORAL | 065055-002 | Dec 1, 2000 | AB | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | |||
| Bausch | DOXYCYCLINE HYCLATE | doxycycline hyclate | CAPSULE, DELAYED RELEASE;ORAL | 065281-002 | Dec 21, 2005 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for doxycycline
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | ⤷ Sign Up | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
EU/EMA Drug Approvals for doxycycline
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pfizer Limited | Doxirobe | Doxycycline | EMEA/V/C/000044 Treatment of periodontal disease in dogs.Periodontal pocket probing depths >=4 mm are evidence of disease that may be responsive to treatment with the Doxirobe Gel. Use of this product as directed should result in attachment level gains, periodontal pocket depth reductions, local antimicrobial effect and improved gingival health. Noticeable improvements in these parameters should be evident within 2-4 weeks following treatment. The response in individual dogs is dependent on the severity of the condition and rigor of adjunctive therapy. |
Withdrawn | no | no | no | 1999-09-16 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for doxycycline
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2004091483 | ⤷ Sign Up | |
| Denmark | 1615622 | ⤷ Sign Up | |
| Canada | 2803922 | FORMULATIONS DE TETRACYCLINES EN DOSE QUOTIDIENNE UNIQUE (ONCE DAILY FORMULATIONS OF TETRACYCLINES) | ⤷ Sign Up |
| Poland | 1615622 | ⤷ Sign Up | |
| Portugal | 1615622 | ⤷ Sign Up | |
| Japan | 2006522162 | ⤷ Sign Up | |
| Canada | 2894736 | FORMULATIONS DE TETRACYCLINES EN DOSE QUOTIDIENNE UNIQUE (ONCE DAILY FORMULATIONS OF TETRACYCLINES) | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.