BOEHRINGER INGELHEIM Company Profile
✉ Email this page to a colleague
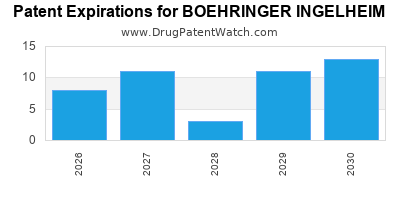
What is the competitive landscape for BOEHRINGER INGELHEIM, and when can generic versions of BOEHRINGER INGELHEIM drugs launch?
BOEHRINGER INGELHEIM has forty-eight approved drugs.
There are sixty-nine US patents protecting BOEHRINGER INGELHEIM drugs.
There are one thousand seven hundred and eighty-two patent family members on BOEHRINGER INGELHEIM drugs in sixty-three countries and two hundred and seventeen supplementary protection certificates in nineteen countries.
Summary for BOEHRINGER INGELHEIM
| International Patents: | 1782 |
| US Patents: | 69 |
| Tradenames: | 37 |
| Ingredients: | 29 |
| NDAs: | 48 |
| Patent Litigation for BOEHRINGER INGELHEIM: | See patent lawsuits for BOEHRINGER INGELHEIM |
| PTAB Cases with BOEHRINGER INGELHEIM as petitioner: | See PTAB cases with BOEHRINGER INGELHEIM as petitioner |
Drugs and US Patents for BOEHRINGER INGELHEIM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | MIRAPEX ER | pramipexole dihydrochloride | TABLET, EXTENDED RELEASE;ORAL | 022421-004 | Feb 19, 2010 | AB | RX | Yes | No | 7,695,734 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Boehringer Ingelheim | SYNJARDY XR | empagliflozin; metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 208658-003 | Dec 9, 2016 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Boehringer Ingelheim | JARDIANCE | empagliflozin | TABLET;ORAL | 204629-002 | Aug 1, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Boehringer Ingelheim | JARDIANCE | empagliflozin | TABLET;ORAL | 204629-002 | Aug 1, 2014 | RX | Yes | Yes | 8,551,957*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for BOEHRINGER INGELHEIM
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Boehringer Ingelheim | TRIJARDY XR | empagliflozin; linagliptin; metformin hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 212614-001 | Jan 27, 2020 | 6,488,962 | ⤷ Try a Trial |
| Boehringer Ingelheim | JENTADUETO | linagliptin; metformin hydrochloride | TABLET;ORAL | 201281-001 | Jan 30, 2012 | 8,119,648 | ⤷ Try a Trial |
| Boehringer Ingelheim | COMBIVENT RESPIMAT | albuterol sulfate; ipratropium bromide | SPRAY, METERED;INHALATION | 021747-001 | Oct 7, 2011 | 6,977,042 | ⤷ Try a Trial |
| Boehringer Ingelheim | STIOLTO RESPIMAT | olodaterol hydrochloride; tiotropium bromide | SPRAY, METERED;INHALATION | 206756-001 | May 21, 2015 | 7,246,615 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for BOEHRINGER INGELHEIM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 2.5 mg/500 mg, 2.5 mg/850 mg, 2.5 mg/1000 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Tablets | 80 mg/12.5 mg and 40 mg/12.5 mg | ➤ Subscribe | 2008-12-31 |
| ➤ Subscribe | Extended-release Capsules | 25 mg and 200 mg | ➤ Subscribe | 2007-02-01 |
| ➤ Subscribe | Tablets | 0.125 mg, 0.5 mg, 1 mg and 1.5 mg | ➤ Subscribe | 2005-06-24 |
| ➤ Subscribe | Capsules | eq. to 75 mg base and 150 mg base | ➤ Subscribe | 2014-10-20 |
| ➤ Subscribe | Tablets | 0.75 mg | ➤ Subscribe | 2008-07-31 |
| ➤ Subscribe | Inhalation Powder Capsules | 18 mcg | ➤ Subscribe | 2018-05-11 |
| ➤ Subscribe | Tablets | 5 mg/500 mg5 mg/1000 mg12.5 mg/500 mg12.5 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Extended-release Tablets | 0.375 mg, 0.75 mg, 1.5 mg, 3 mg and 4.5 mg | ➤ Subscribe | 2010-06-01 |
| ➤ Subscribe | Extended-release Tablets | 5 mg/1000 mg10 mg/1000 mg12.5 mg/1000 mg25 mg/1000 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 30 mg and 40 mg | ➤ Subscribe | 2017-07-12 |
| ➤ Subscribe | Tablets | 10 mg and 25 mg | ➤ Subscribe | 2018-08-01 |
| ➤ Subscribe | Tablets | 20 mg, 40 mg and 80 mg | ➤ Subscribe | 2006-12-26 |
| ➤ Subscribe | Tablets | 80 mg/25 mg | ➤ Subscribe | 2009-02-27 |
| ➤ Subscribe | Capsules | 100 mg and 150 mg | ➤ Subscribe | 2018-10-15 |
| ➤ Subscribe | Tablets | 0.25 mg | ➤ Subscribe | 2005-05-27 |
| ➤ Subscribe | Capsules | eq. to 110 mg base | ➤ Subscribe | 2015-12-15 |
| ➤ Subscribe | Oral Suspension | 7.5 mg/5 mL | ➤ Subscribe | 2009-12-17 |
| ➤ Subscribe | Extended-releaseTablets | 2.5 mg/1000 mg 5 mg/1000 mg | ➤ Subscribe | 2018-03-28 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2015-05-04 |
| ➤ Subscribe | Extended-release Tablets | 2.25 mg and 3.75 mg | ➤ Subscribe | 2011-07-26 |
| ➤ Subscribe | Extended-release Tablets | 400 mg | ➤ Subscribe | 2013-06-21 |
| ➤ Subscribe | Tablets | 10 mg/5 mg and 25 mg/5 mg | ➤ Subscribe | 2018-08-01 |
International Patents for BOEHRINGER INGELHEIM Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2539285 | ⤷ Try a Trial |
| Canada | 2787566 | ⤷ Try a Trial |
| Bulgaria | 106587 | ⤷ Try a Trial |
| Japan | 2012072187 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for BOEHRINGER INGELHEIM Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2525812 | PA2017021 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: IDARUCIZUMABAS; REGISTRATION NO/DATE: EU/1/15/1056/001 20151120 |
| 1345910 | SPC/GB14/015 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: AFATINIB, OPTIONALLY IN THE FORM OF A PHYSIOLOGICALLY ACCEPTABLE SALT, IN PARTICULAR A SALT WITH MALEIC ACID; REGISTERED: UK EU/1/13/879/001 20130927; UK EU/1/13/879/002 20130927; UK EU/1/13/879/003 20130927; UK EU/1/13/879/004 20130927; UK EU/1/13/879/005 20130927; UK EU/1/13/879/006 20130927; UK EU/1/13/879/007 20130927; UK EU/1/13/879/008 20130927; UK EU/1/13/879/009 20130927; UK EU/1/13/879/010 20130927; UK EU/1/13/879/011 20130927; UK EU/1/13/879/012 20130927 |
| 1345910 | 6/2014 | Austria | ⤷ Try a Trial | PRODUCT NAME: AFATINIB, DEREN TAUTOMERE, DEREN STEREOISOMERE UND DEREN SALZE, PHYSIOLOGISCH VERTRAEGLICHE SALZE MIT ANORGANISCHEN ODER ORGANISCHEN SAEUREN ODER BASEN, BEVORZUGT DEREN MALEATSALZE, VORZUGSWEISE DEREN DIMALEATSALZE; REGISTRATION NO/DATE: EU/1/13/879 20130925 |
| 2525812 | CR 2017 00031 | Denmark | ⤷ Try a Trial | PRODUCT NAME: IDARUCIZUMAB; REG. NO/DATE: EU/1/15/1056 20151124 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.