GALDERMA LABS LP Company Profile
✉ Email this page to a colleague
What is the competitive landscape for GALDERMA LABS LP, and what generic alternatives to GALDERMA LABS LP drugs are available?
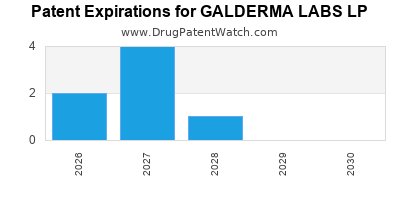
GALDERMA LABS LP has twenty-six approved drugs.
There are fifty-eight US patents protecting GALDERMA LABS LP drugs.
There are four hundred and four patent family members on GALDERMA LABS LP drugs in thirty-seven countries and one hundred and seventy-eight supplementary protection certificates in eighteen countries.
Summary for GALDERMA LABS LP
| International Patents: | 404 |
| US Patents: | 58 |
| Tradenames: | 18 |
| Ingredients: | 16 |
| NDAs: | 26 |
Drugs and US Patents for GALDERMA LABS LP
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | SOOLANTRA | ivermectin | CREAM;TOPICAL | 206255-001 | Dec 19, 2014 | AB | RX | Yes | Yes | 8,415,311 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Galderma Labs Lp | DIFFERIN | adapalene | SOLUTION;TOPICAL | 020338-001 | May 31, 1996 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Galderma Labs Lp | MIRVASO | brimonidine tartrate | GEL;TOPICAL | 204708-001 | Aug 23, 2013 | AB | RX | Yes | Yes | 8,163,725 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Galderma Labs Lp | METROCREAM | metronidazole | CREAM;TOPICAL | 020531-001 | Sep 20, 1995 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Galderma Labs Lp | TRI-LUMA | fluocinolone acetonide; hydroquinone; tretinoin | CREAM;TOPICAL | 021112-001 | Jan 18, 2002 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Galderma Labs Lp | EPSOLAY | benzoyl peroxide | CREAM;TOPICAL | 214510-001 | Apr 22, 2022 | RX | Yes | Yes | 10,945,987 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Galderma Labs Lp | EPIDUO | adapalene; benzoyl peroxide | GEL;TOPICAL | 022320-001 | Dec 8, 2008 | AB | RX | Yes | Yes | 7,820,186 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for GALDERMA LABS LP
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | 5,919,775 | ⤷ Try a Trial |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | 7,211,267 | ⤷ Try a Trial |
| Galderma Labs Lp | CLOBEX | clobetasol propionate | SPRAY;TOPICAL | 021835-001 | Oct 27, 2005 | 5,990,100 | ⤷ Try a Trial |
| Galderma Labs Lp | ORACEA | doxycycline | CAPSULE;ORAL | 050805-001 | May 26, 2006 | 7,232,572 | ⤷ Try a Trial |
| Galderma Labs Lp | METROGEL | metronidazole | GEL;TOPICAL | 019737-001 | Nov 22, 1988 | 4,837,378 | ⤷ Try a Trial |
| Galderma Labs Lp | TRI-LUMA | fluocinolone acetonide; hydroquinone; tretinoin | CREAM;TOPICAL | 021112-001 | Jan 18, 2002 | 7,939,516 | ⤷ Try a Trial |
| Galderma Labs Lp | DIFFERIN | adapalene | CREAM;TOPICAL | 020748-001 | May 26, 2000 | RE34440 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for GALDERMA LABS LP drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Topical Gel | 0.30% | ➤ Subscribe | 2009-09-15 |
| ➤ Subscribe | Gel | 0.1%/2.5% | ➤ Subscribe | 2011-12-30 |
| ➤ Subscribe | Topical Gel | 0.33% | ➤ Subscribe | 2014-12-15 |
| ➤ Subscribe | Spray | 0.05% | ➤ Subscribe | 2008-09-29 |
| ➤ Subscribe | Delayed-release Capsules | 40 mg | ➤ Subscribe | 2008-12-11 |
| ➤ Subscribe | Cream | 1% | ➤ Subscribe | 2016-12-30 |
| ➤ Subscribe | Topical Gel | 1% | ➤ Subscribe | 2008-10-21 |
| ➤ Subscribe | Lotion | 0.05% | ➤ Subscribe | 2006-03-27 |
| ➤ Subscribe | Topical Shampoo | 0.05% | ➤ Subscribe | 2008-01-09 |
International Patents for GALDERMA LABS LP Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Russian Federation | 2009104949 | ⤷ Try a Trial |
| Portugal | 1485080 | ⤷ Try a Trial |
| Canada | 2824612 | ⤷ Try a Trial |
| South Korea | 20170018974 | ⤷ Try a Trial |
| Poland | 2444068 | ⤷ Try a Trial |
| Poland | 2815748 | ⤷ Try a Trial |
| Argentina | 057866 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for GALDERMA LABS LP Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1458369 | CA 2008 00029 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ADAPALEN, BENZOYLPEROXID |
| 1870100 | 122012000047 | Germany | ⤷ Try a Trial | PRODUCT NAME: DABIGATRAN ETEXILAT MESILAT = 3-((2-((4-(HEXYLOXYCARBONYLAMINO-IMINO-METHYL)-PHENYLAMINO)-METHYL)-1-METHYL-1H-BENZIMIDAZOL-5-CARBONYL)-PYRIDIN-2-YL-AMINO)-PROPIONSAEURE-ETHYLESTER-METHANSULFONAT; REGISTRATION NO/DATE: EU/1/08/442/001-008 20080318 |
| 1948158 | 300810 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS HET SACUBITRIL VALSARTAN NATRIUMZOUTCOMPLEX, IE. TRINATRIUM (3-((LS,3R)-1-BIFENYL-4-YLMETHYL-3-ETHOXYCARBONYL-1-BUTYLCARBAMOYL)PROPIONAAT-(S)-3'-METHYL-2'-(PENTANOYL(2"-(TETRAZOL-5-YLAAT)BIFENYL-4'-YLMETHYL)AMINO)BUTYRAAT) HEMIPENTAHYDRAAT; REGISTRATION NO/DATE: EU/1/15/1058 20151123 |
| 2233112 | 132014902285293 | Italy | ⤷ Try a Trial | PRODUCT NAME: FLUOCINOLONE ACETONIDE(ILUVIEN); AUTHORISATION NUMBER(S) AND DATE(S): 042616019, 20140530;PL27813/0001, 20120504 |
| 1831149 | 301042 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: TRIFAROTEEN, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; NATIONAL REGISTRATION NO/DATE: RVG 124058 20200310; FIRST REGISTRATION: GB PL 10590/0071 20200113 |
| 2340828 | 122021000003 | Germany | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, ALS SACUBITRIL VALSARTAN NATRIUM SALZKOMPLEX, D.H. (((S)-N-VALERYL-N-((2'-(1H-TETRAZOL-5-YL)-BIPHENYL-4-YL)-METHYL)-VALIN) ((2R,4S)-5-BIPHENYL-4-YL-4-(3-CARBOXY-PROPIONYLAMINO)-2-METHYL-PENTANSAEUREETHYLESTER))NA 3 * X H 2 0, WORIN X 0 BIS 3 IST; REGISTRATION NO/DATE: EU/1/15/1058 20151119 |
| 1631293 | 92462 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE ET SES SELS PHARMACEUTIQUES POUR L UTILISATION COMME MEDICAMENT POUR LE TRAITEMENT DES ROUGEURS INDUITES PAR LA ROSACEA.FIRST REGISTRATION: 20140225 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.