Array Biopharma Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ARRAY BIOPHARMA INC, and when can generic versions of ARRAY BIOPHARMA INC drugs launch?
ARRAY BIOPHARMA INC has two approved drugs.
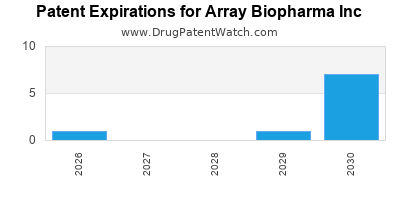
There are seventeen US patents protecting ARRAY BIOPHARMA INC drugs.
There are three hundred and seven patent family members on ARRAY BIOPHARMA INC drugs in fifty-seven countries and forty-five supplementary protection certificates in nineteen countries.
Summary for Array Biopharma Inc
| International Patents: | 307 |
| US Patents: | 17 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for Array Biopharma Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-002 | Jun 27, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-001 | Jun 27, 2018 | DISCN | Yes | No | 9,850,229 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-002 | Jun 27, 2018 | RX | Yes | Yes | 9,763,941 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Array Biopharma Inc | BRAFTOVI | encorafenib | CAPSULE;ORAL | 210496-001 | Jun 27, 2018 | DISCN | Yes | No | 9,593,100 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Array Biopharma Inc | MEKTOVI | binimetinib | TABLET;ORAL | 210498-001 | Jun 27, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Array Biopharma Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Array Biopharma Inc | MEKTOVI | binimetinib | TABLET;ORAL | 210498-001 | Jun 27, 2018 | 8,513,293 | ⤷ Try a Trial |
| Array Biopharma Inc | MEKTOVI | binimetinib | TABLET;ORAL | 210498-001 | Jun 27, 2018 | 8,178,693 | ⤷ Try a Trial |
| Array Biopharma Inc | MEKTOVI | binimetinib | TABLET;ORAL | 210498-001 | Jun 27, 2018 | 8,193,229 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Array Biopharma Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Iceland | 8960 | ⤷ Try a Trial |
| Russian Federation | 2004127926 | ⤷ Try a Trial |
| San Marino | T201700036 | ⤷ Try a Trial |
| Colombia | 7200273 | ⤷ Try a Trial |
| Eurasian Patent Organization | 201500175 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Array Biopharma Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2470526 | CR 2019 00012 | Denmark | ⤷ Try a Trial | PRODUCT NAME: ENCORAFENIB ELLER ET FARMACEUTISK ACCEPTABELT SALT ELLER SOLVAT DERAF; REG. NO/DATE: EU/1/18/1314 20180924 |
| 2470526 | 1990012-5 | Sweden | ⤷ Try a Trial | PRODUCT NAME: ENCORAFENIB OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEROF; REG. NO/DATE: EU/1/18/1314 20180924 |
| 1482932 | SPC/GB19/007 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: BINIMETINIB OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF; REGISTERED: UK EU/1/18/1315/001-002 20180920 |
| 2470526 | LUC00101 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: ENCORAFENIB SOUS TOUTES SES FORMES TELLES QUE PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/18/1314 20180924 |
| 1482932 | 2019C/510 | Belgium | ⤷ Try a Trial | PRODUCT NAME: BINIMETINIB SOUS TOUTES SES FORMES PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/18/1315 20180924 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.