Azurity Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AZURITY, and when can generic versions of AZURITY drugs launch?
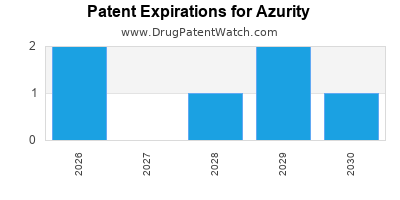
AZURITY has twenty-three approved drugs.
There are seventy-eight US patents protecting AZURITY drugs.
There are three hundred and thirty-five patent family members on AZURITY drugs in forty-eight countries and forty-one supplementary protection certificates in fifteen countries.
Summary for Azurity
| International Patents: | 335 |
| US Patents: | 78 |
| Tradenames: | 23 |
| Ingredients: | 22 |
| NDAs: | 23 |
| Patent Litigation for Azurity: | See patent lawsuits for Azurity |
Drugs and US Patents for Azurity
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Azurity | FIRVANQ KIT | vancomycin hydrochloride | FOR SOLUTION;ORAL | 208910-001 | Jan 26, 2018 | AB | RX | Yes | Yes | 10,959,948 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Azurity | FIRVANQ KIT | vancomycin hydrochloride | FOR SOLUTION;ORAL | 208910-001 | Jan 26, 2018 | AB | RX | Yes | Yes | 10,959,949 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Azurity | EPRONTIA | topiramate | SOLUTION;ORAL | 214679-001 | Nov 5, 2021 | RX | Yes | Yes | 11,433,046 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Azurity | ZONISADE | zonisamide | SUSPENSION;ORAL | 214273-001 | Jul 15, 2022 | RX | Yes | Yes | 11,529,333 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Azurity | FIRVANQ KIT | vancomycin hydrochloride | FOR SOLUTION;ORAL | 208910-002 | Jan 26, 2018 | AB | RX | Yes | Yes | 10,959,949 | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Azurity | SOTYLIZE | sotalol hydrochloride | SOLUTION;ORAL | 205108-001 | Oct 22, 2014 | RX | Yes | Yes | 11,850,222 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Azurity
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Azurity | EDARBI | azilsartan kamedoxomil | TABLET;ORAL | 200796-001 | Feb 25, 2011 | 5,583,141 | ⤷ Try a Trial |
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-004 | Jan 30, 2019 | 10,130,580 | ⤷ Try a Trial |
| Azurity | EDARBI | azilsartan kamedoxomil | TABLET;ORAL | 200796-002 | Feb 25, 2011 | 5,583,141 | ⤷ Try a Trial |
| Azurity | EVEKEO ODT | amphetamine sulfate | TABLET, ORALLY DISINTEGRATING;ORAL | 209905-005 | Apr 16, 2021 | 10,130,580 | ⤷ Try a Trial |
| Azurity | GLIADEL | carmustine | IMPLANT;INTRACRANIAL | 020637-001 | Sep 23, 1996 | 5,179,189 | ⤷ Try a Trial |
| Azurity | EDARBYCLOR | azilsartan kamedoxomil; chlorthalidone | TABLET;ORAL | 202331-001 | Dec 20, 2011 | 5,736,555 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for AZURITY drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Oral Solution | 1 mg/mL | ➤ Subscribe | 2018-08-31 |
International Patents for Azurity Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Brazil | PI0414481 | ⤷ Try a Trial |
| Hungary | 0400167 | ⤷ Try a Trial |
| Mexico | PA06004088 | ⤷ Try a Trial |
| Russian Federation | 2377234 | ⤷ Try a Trial |
| South Africa | 201100871 | ⤷ Try a Trial |
| Denmark | 2275401 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Azurity Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1718641 | CA 2012 00013 | Denmark | ⤷ Try a Trial | |
| 1718641 | C20120004 00052 | Estonia | ⤷ Try a Trial | PRODUCT NAME: EDARBI - ASILSARTAANMEDOKSOMIIL;REG NO/DATE: C(2011) 9280 FINAL 07.12.2011 |
| 0443983 | 2007C/043 | Belgium | ⤷ Try a Trial | PRODUCT NAME: AMLODIPINE ET VALSARTAN; NATL. REGISTRATION NO/DATE: EU/1/06/370/001 20070118; FIRST REGISTRATION: CH 57771 20061222 |
| 1507558 | C300528 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE VAN ALISKIREN OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, AMLODIPINE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN EN HYDROCHLOORTHIAZIDE OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; NATL. REGISTRATION NO/DATE: EU/1/11/730/001-060 20111122; FIRST REGISTRATION: CH 61678 01-05 20110705 |
| 2119715 | 300802 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: AZILSARTAN MEDOXOMIL EN CHLOORTALIDON; NATIONAL REGISTRATION NO/DATE: RVG116387 20151125; FIRST REGISTRATION: CH 6314502 20141028 |
| 1915993 | C300625 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMBINATIE BEVATTENDE ALISKIREN, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN, EN AMLODIPINE, OF EEN FARMACEUATISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/11/686/001-056 20110414 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.