Mayne Pharma Company Profile
✉ Email this page to a colleague
What is the competitive landscape for MAYNE PHARMA, and what generic alternatives to MAYNE PHARMA drugs are available?
MAYNE PHARMA has fifteen approved drugs.
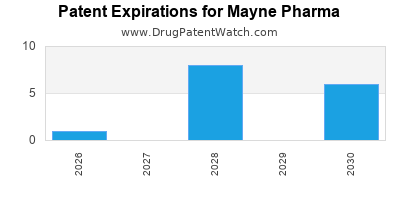
There are seventy-eight US patents protecting MAYNE PHARMA drugs. There is one tentative approval on MAYNE PHARMA drugs.
There are four hundred and twenty-six patent family members on MAYNE PHARMA drugs in forty-nine countries and sixty supplementary protection certificates in fourteen countries.
Summary for Mayne Pharma
| International Patents: | 426 |
| US Patents: | 78 |
| Tradenames: | 18 |
| Ingredients: | 14 |
| NDAs: | 15 |
Drugs and US Patents for Mayne Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mayne Pharma | FABIOR | tazarotene | AEROSOL, FOAM;TOPICAL | 202428-001 | May 11, 2012 | RX | Yes | Yes | 8,808,716 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Mayne Pharma | LEXETTE | halobetasol propionate | AEROSOL, FOAM;TOPICAL | 210566-001 | May 24, 2018 | AB | RX | Yes | Yes | 10,857,159*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | ||
| Mayne Pharma | BIJUVA | estradiol; progesterone | CAPSULE;ORAL | 210132-001 | Oct 28, 2018 | RX | Yes | Yes | 11,865,179 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Mayne Pharma
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Mayne Pharma | DORYX MPC | doxycycline hyclate | TABLET, DELAYED RELEASE;ORAL | 050795-008 | May 20, 2016 | 6,958,161 | ⤷ Try a Trial |
| Mayne Pharma | DORYX | doxycycline hyclate | TABLET, DELAYED RELEASE;ORAL | 050795-001 | May 6, 2005 | 6,958,161 | ⤷ Try a Trial |
| Mayne Pharma | TOLSURA | itraconazole | CAPSULE;ORAL | 208901-001 | Dec 11, 2018 | 8,771,739 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for MAYNE PHARMA drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Delayed-release Tablets | 200 mg | ➤ Subscribe | 2014-05-19 |
| ➤ Subscribe | Delayed-release Tablets | 50 mg | ➤ Subscribe | 2015-11-05 |
| ➤ Subscribe | Delayed-release Tablets | 200 mg | ➤ Subscribe | 2013-08-12 |
| ➤ Subscribe | Delayed-release Tablets | 80 mg | ➤ Subscribe | 2015-07-01 |
| ➤ Subscribe | Delayed-release Tablets | 60 mg and 120 mg | ➤ Subscribe | 2017-09-28 |
| ➤ Subscribe | Delayed-release Tablets | 150 mg | ➤ Subscribe | 2008-12-19 |
International Patents for Mayne Pharma Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Mexico | 366829 | ⤷ Try a Trial |
| Argentina | 099872 | ⤷ Try a Trial |
| Hong Kong | 1078780 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Mayne Pharma Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0136011 | 99C0003 | Belgium | ⤷ Try a Trial | PRODUCT NAME: ESTRADIOL AND NORETHISTERONE; FIRST REGISTRATION NO/DATE: 403 IS 106 F3 19980928; FIRST REGISTRATION: SE 14007 19980306 |
| 0836511 | SPC/GB06/022 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: FENTANYL HYDROCHLORIDE; REGISTERED: UK EU/1/05/326/001 20060124 |
| 2782584 | 301153 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: COMPOSITION CONTAINING BOTH ESTRADIOL (17SS-ESTRADIOL), OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT, HYDRATE OR SOLVATE THEREOF (INCLUDING IN HEMIHYDRATE FORM), AND PROGESTERONE; NATIONAL REGISTRATION NO/DATE: RVG 125821 20210611; FIRST REGISTRATION: BE BE582231 20210406 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.