Novartis Pharm Company Profile
✉ Email this page to a colleague
What is the competitive landscape for NOVARTIS PHARM, and when can generic versions of NOVARTIS PHARM drugs launch?
NOVARTIS PHARM has five approved drugs.
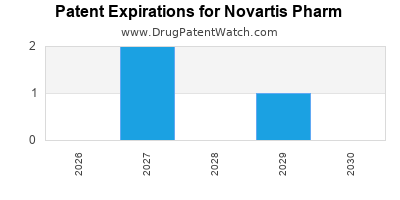
There are fourteen US patents protecting NOVARTIS PHARM drugs.
There are three hundred and fifty-one patent family members on NOVARTIS PHARM drugs in fifty-four countries and ninety-two supplementary protection certificates in seventeen countries.
Summary for Novartis Pharm
| International Patents: | 351 |
| US Patents: | 14 |
| Tradenames: | 5 |
| Ingredients: | 5 |
| NDAs: | 5 |
| Patent Litigation for Novartis Pharm: | See patent lawsuits for Novartis Pharm |
Drugs and US Patents for Novartis Pharm
Expired US Patents for Novartis Pharm
Paragraph IV (Patent) Challenges for NOVARTIS PHARM drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 2.5 mg | ➤ Subscribe | 2006-03-02 |
| ➤ Subscribe | Tablets | 180 mg | ➤ Subscribe | 2015-10-23 |
| ➤ Subscribe | Tablets for Oral Suspension | 2 mg, 3 mg and 5 mg | ➤ Subscribe | 2016-12-30 |
| ➤ Subscribe | Tablets | 90 mg and 360 mg | ➤ Subscribe | 2015-10-19 |
| ➤ Subscribe | Tablets | 180 mg | ➤ Subscribe | 2016-04-28 |
International Patents for Novartis Pharm Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Norway | 337288 | ⤷ Try a Trial |
| Hong Kong | 1212242 | ⤷ Try a Trial |
| Japan | 5572388 | ⤷ Try a Trial |
| China | 103936743 | ⤷ Try a Trial |
| Colombia | 2017000586 | ⤷ Try a Trial |
| Poland | 2099447 | ⤷ Try a Trial |
| Japan | 7002587 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Novartis Pharm Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1467728 | CA 2016 00022 | Denmark | ⤷ Try a Trial | PRODUCT NAME: SACUBITRIL/VALSARTAN, HERUNDER FARMACEUTISK ACCEPTABLE SALTE DERAF; REG. NO/DATE: EU/1/15/1058 (C(2015)8288) 20151123 |
| 2269603 | 122015000094 | Germany | ⤷ Try a Trial | PRODUCT NAME: EVEROLIMUS ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/09/538/001 20120723 |
| 3143995 | 122019000051 | Germany | ⤷ Try a Trial | PRODUCT NAME: EVEROLIMUS ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ DAVON; REGISTRATION NO/DATE: EU/1/09/538/001 EU/1/09/538/003 EU/1/09/538/004 EU/1/09/538/006 EU/1/09/538/007 EU/1/09/538/008 EU/1/09/538/009 EU/1/09/538/010 20160526 |
| 3351246 | C03351246/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: EVEROLIMUS; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 59174 27.02.2013 |
| 3143995 | C03143995/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: EVEROLIMUS; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 59174 14.11.2016 |
| 0663916 | CA 2004 00020 | Denmark | ⤷ Try a Trial | PRODUCT NAME: EVEROLIMUS |
| 0914118 | SPC/GB07/002 | United Kingdom | ⤷ Try a Trial | SUPPLEMENTARY PROTECTION CERTIFICATE NO SPC/GB07/002 GRANTED TO NOVARTIS AG IN RESPECT OF THE PRODUCT DEFERASIROX AND PHARMACEUTICALLY ACCEPTABLE SALTS THEREOF, ESPECIALLY 4-(3,5-BIS(2-HYDROXYPHENYL)-(1,2,4) TRIAZOL-L-YL) BENZOIC ACID, THE GRANT OF WHICH WAS ADVERTISED IN JOURNAL NO 6179 DATED 24 OCTOBER 2007 HAS HAD ITS MAXIMUM PERIOD OF DURATION CORRECTED, SUBJECT TO THE PAYMENT OF THE PRESCRIBED FEES IT WILL EXPIRE ON 30 AUGUST 2021. |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.