Vertex Pharms Company Profile
✉ Email this page to a colleague
What is the competitive landscape for VERTEX PHARMS, and when can generic versions of VERTEX PHARMS drugs launch?
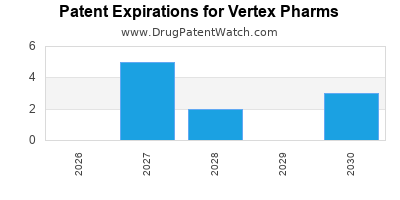
VERTEX PHARMS has eight approved drugs.
There are forty-nine US patents protecting VERTEX PHARMS drugs.
There are seven hundred and eighty-one patent family members on VERTEX PHARMS drugs in fifty-four countries and eighty-four supplementary protection certificates in nineteen countries.
Drugs and US Patents for Vertex Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vertex Pharms Inc | SYMDEKO (COPACKAGED) | ivacaftor; ivacaftor, tezacaftor | TABLET;ORAL | 210491-002 | Jun 21, 2019 | RX | Yes | No | 9,012,496 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Vertex Pharms Inc | ORKAMBI | ivacaftor; lumacaftor | TABLET;ORAL | 206038-001 | Jul 2, 2015 | RX | Yes | Yes | 10,646,481 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Vertex Pharms Inc | TRIKAFTA (COPACKAGED) | elexacaftor, ivacaftor, tezacaftor; ivacaftor | TABLET;ORAL | 212273-001 | Oct 21, 2019 | RX | Yes | Yes | 11,179,367 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Vertex Pharms Inc | ORKAMBI | ivacaftor; lumacaftor | GRANULE;ORAL | 211358-001 | Aug 7, 2018 | RX | Yes | No | 10,597,384 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Vertex Pharms Inc | ORKAMBI | ivacaftor; lumacaftor | GRANULE;ORAL | 211358-002 | Aug 7, 2018 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Vertex Pharms
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Vertex Pharms | INCIVEK | telaprevir | TABLET;ORAL | 201917-001 | May 23, 2011 | 8,529,882 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Premature patent expirations for VERTEX PHARMS
Expiration due to failure to pay maintenance fee
| Patent Number | Expiration Date |
|---|---|
| ⤷ Try a Trial | ⤷ Try a Trial |
International Patents for Vertex Pharms Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| European Patent Office | 1958956 | ⤷ Try a Trial |
| Israel | 221828 | ⤷ Try a Trial |
| China | 103641765 | ⤷ Try a Trial |
| European Patent Office | 3551622 | ⤷ Try a Trial |
| European Patent Office | 1993360 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Vertex Pharms Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2404919 | 1690018-5 | Sweden | ⤷ Try a Trial | PRODUCT NAME: 3-6-1-(2,2-DIFLUORO-1,3-BENZODIOXOL-5- YL)CYCLOPROPANECARBONYLAMINO-3-METHYLPYRIDIN-2-YLBENZOIC ACID, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, OR AN ESTER THEREOF.; REG. NO/DATE: EU/1/15/1059 20151124 |
| 1320540 | 2012/009 | Ireland | ⤷ Try a Trial | PRODUCT NAME: TELAPREVIR, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT AND/OR SOLVATE THEREOF OR SOLVATES OF SUCH SALTS; REGISTRATION NO/DATE: EU/1/11/720/001 20110919 |
| 1320540 | C300518 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: TELAPREVIR, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT EN/OF SOLVAAT DAARVAN, OF SOLVATEN VAN DERGELIJKE ZOUTEN; REGISTRATION NO/DATE: EU/1/11/720/001 2011190919 |
| 2826776 | C20210011 00330 | Estonia | ⤷ Try a Trial | PRODUCT NAME: TESAKAFTOOR/IVAKAFTOOR;REG NO/DATE: EU/1/18/1306 06.11.2018 |
| 2395002 | 300812 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: 3-(6-((1-(2,2-DIFLUOR-1,3-BENZODIOXOL-5-YL)CYCLOPROPAANCARBONYL)AMINO)-3-METHYLPYRIDIN-2-YL)BENZOEZUUR, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT DAARVAN; REGISTRATION NO/DATE: EU/1/15/1059 20151124 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.