BACLOFEN Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Baclofen, and when can generic versions of Baclofen launch?
Baclofen is a drug marketed by Acic Pharms, Amneal, Maia Pharms Inc, Mylan Labs Ltd, Rubicon, Ani Pharms, Appco, Aurobindo Pharma Ltd, Beximco Pharms Usa, Eywa Pharma, Graviti Pharms, Impax, Innogenix, Ivax Sub Teva Pharms, Lannett Co Inc, Mankind Pharma, Micro Labs, Mylan, Northstar Hlthcare, Oxford Pharms, Rising, Sun Pharm Inds Inc, Teva, Unichem, Upsher Smith Labs, Usl Pharma, Vintage Pharms, Watson Labs, and Zydus. and is included in forty-one NDAs.
The generic ingredient in BACLOFEN is baclofen. There are twenty-one drug master file entries for this compound. Sixty-four suppliers are listed for this compound. Additional details are available on the baclofen profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Baclofen
A generic version of BACLOFEN was approved as baclofen by IVAX SUB TEVA PHARMS on July 21st, 1988.
Summary for BACLOFEN
| US Patents: | 0 |
| Applicants: | 29 |
| NDAs: | 41 |
| Finished Product Suppliers / Packagers: | 54 |
| Raw Ingredient (Bulk) Api Vendors: | 149 |
| Clinical Trials: | 121 |
| Patent Applications: | 4,743 |
| Formulation / Manufacturing: | see details |
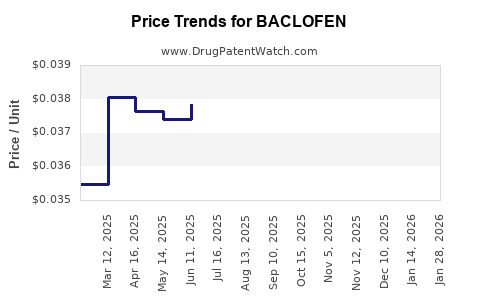
| Drug Prices: | Drug price information for BACLOFEN |
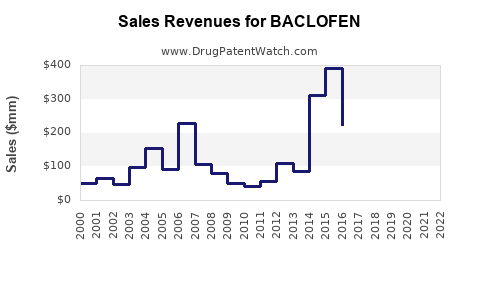
| Drug Sales Revenues: | Drug sales revenues for BACLOFEN |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BACLOFEN |
| What excipients (inactive ingredients) are in BACLOFEN? | BACLOFEN excipients list |
| DailyMed Link: | BACLOFEN at DailyMed |
Recent Clinical Trials for BACLOFEN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Kuwait University | Early Phase 1 |
| Marmara University | N/A |
| European University of Lefke | N/A |
Pharmacology for BACLOFEN
| Drug Class | gamma-Aminobutyric Acid-ergic Agonist |
| Mechanism of Action | GABA A Agonists GABA B Agonists |
Medical Subject Heading (MeSH) Categories for BACLOFEN
Anatomical Therapeutic Chemical (ATC) Classes for BACLOFEN
US Patents and Regulatory Information for BACLOFEN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Watson Labs | BACLOFEN | baclofen | TABLET;ORAL | 072824-001 | Sep 18, 1991 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mankind Pharma | BACLOFEN | baclofen | TABLET;ORAL | 215885-002 | Jan 25, 2022 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Innogenix | BACLOFEN | baclofen | TABLET;ORAL | 212378-003 | Apr 30, 2021 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Northstar Hlthcare | BACLOFEN | baclofen | TABLET;ORAL | 078401-001 | Sep 18, 2009 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Sun Pharm Inds Inc | BACLOFEN | baclofen | TABLET;ORAL | 077862-002 | Aug 14, 2006 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


