BYVALSON Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Byvalson, and when can generic versions of Byvalson launch?
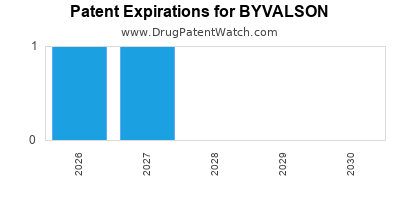
Byvalson is a drug marketed by Abbvie and is included in one NDA. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has thirty-seven patent family members in fifteen countries.
The generic ingredient in BYVALSON is nebivolol hydrochloride; valsartan. There are fourteen drug master file entries for this compound. Additional details are available on the nebivolol hydrochloride; valsartan profile page.
DrugPatentWatch® Generic Entry Outlook for Byvalson
There is one Paragraph IV patent challenge for this drug. This may lead to patent invalidation or a license for generic production.
Indicators of Generic Entry
Summary for BYVALSON
| International Patents: | 37 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 1 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for BYVALSON |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for BYVALSON |
| DailyMed Link: | BYVALSON at DailyMed |

Anatomical Therapeutic Chemical (ATC) Classes for BYVALSON
Paragraph IV (Patent) Challenges for BYVALSON
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| BYVALSON | Tablets | nebivolol hydrochloride; valsartan | 5 mg/80 mg | 206302 | 1 | 2017-06-09 |
US Patents and Regulatory Information for BYVALSON
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Abbvie | BYVALSON | nebivolol hydrochloride; valsartan | TABLET;ORAL | 206302-001 | Jun 3, 2016 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | ⤷ Sign Up | ||||
| Abbvie | BYVALSON | nebivolol hydrochloride; valsartan | TABLET;ORAL | 206302-001 | Jun 3, 2016 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for BYVALSON
When does loss-of-exclusivity occur for BYVALSON?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 15
Estimated Expiration: ⤷ Sign Up
Patent: 96
Estimated Expiration: ⤷ Sign Up
Australia
Patent: 05249514
Estimated Expiration: ⤷ Sign Up
Patent: 05332300
Estimated Expiration: ⤷ Sign Up
Austria
Patent: 09627
Estimated Expiration: ⤷ Sign Up
Canada
Patent: 69881
Estimated Expiration: ⤷ Sign Up
Patent: 10694
Estimated Expiration: ⤷ Sign Up
China
Patent: 1022791
Estimated Expiration: ⤷ Sign Up
Patent: 1242827
Estimated Expiration: ⤷ Sign Up
Patent: 2188708
Estimated Expiration: ⤷ Sign Up
European Patent Office
Patent: 53418
Estimated Expiration: ⤷ Sign Up
Patent: 90691
Estimated Expiration: ⤷ Sign Up
Patent: 82711
Estimated Expiration: ⤷ Sign Up
Patent: 74658
Estimated Expiration: ⤷ Sign Up
Patent: 08015
Estimated Expiration: ⤷ Sign Up
Hong Kong
Patent: 04570
Estimated Expiration: ⤷ Sign Up
Israel
Patent: 9999
Estimated Expiration: ⤷ Sign Up
Patent: 7552
Estimated Expiration: ⤷ Sign Up
Japan
Patent: 08545742
Estimated Expiration: ⤷ Sign Up
New Zealand
Patent: 1910
Estimated Expiration: ⤷ Sign Up
Patent: 3759
Estimated Expiration: ⤷ Sign Up
Russian Federation
Patent: 03037
Estimated Expiration: ⤷ Sign Up
Patent: 41653
Estimated Expiration: ⤷ Sign Up
Patent: 93335
Estimated Expiration: ⤷ Sign Up
Patent: 06147220
Estimated Expiration: ⤷ Sign Up
Patent: 07148414
Estimated Expiration: ⤷ Sign Up
Patent: 10130950
Estimated Expiration: ⤷ Sign Up
Singapore
Patent: 2255
Estimated Expiration: ⤷ Sign Up
Patent: 2734
Estimated Expiration: ⤷ Sign Up
South Africa
Patent: 0610708
Estimated Expiration: ⤷ Sign Up
Patent: 0710355
Estimated Expiration: ⤷ Sign Up
South Korea
Patent: 1236166
Estimated Expiration: ⤷ Sign Up
Patent: 1310037
Estimated Expiration: ⤷ Sign Up
Patent: 070044833
Estimated Expiration: ⤷ Sign Up
Patent: 080025699
Estimated Expiration: ⤷ Sign Up
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering BYVALSON around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| China | 101242827 | Compositions comprising nebivolol | ⤷ Sign Up |
| Canada | 2610694 | COMPOSITIONS A BASE DE NEBIVOLOL (COMPOSITIONS COMRISING NEBIVOLOL) | ⤷ Sign Up |
| European Patent Office | 1753418 | COMPOSITIONS COMPRENANT DU NÉBIVOLOL (COMPOSITIONS COMPRISING NEBIVOLOL) | ⤷ Sign Up |
| European Patent Office | 2174658 | Compositions comportant du nebivolol (Compositions comprising nebivolol) | ⤷ Sign Up |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for BYVALSON
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0334429 | SPC/GB96/048 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: NEBIVOLOL, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR HYDRATE; REGISTERED: NL RVG/19317 19951018; UK 00242/0309 19960509 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


