CIMETIDINE Drug Patent Profile
✉ Email this page to a colleague
When do Cimetidine patents expire, and what generic alternatives are available?
Cimetidine is a drug marketed by Apotex, Chartwell Molecules, Chartwell Rx, Contract Pharmacal, Cycle, Hikma, Ivax Sub Teva Pharms, L Perrigo Co, Mylan, Novitium Pharma, Perrigo, Pliva, Sandoz, Teva, Upsher Smith Labs, Watson Labs Inc, Watson Labs Teva, Dava Pharms Inc, Hospira, Luitpold, Teva Parenteral, Vintage Pharms Llc, Ani Pharms, Chartwell Molecular, G And W Labs Inc, Pai Holdings Pharm, Pharm Assoc, and Pharmobedient Cnsltg. and is included in forty-nine NDAs.
The generic ingredient in CIMETIDINE is cimetidine hydrochloride. There are twenty-five drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the cimetidine hydrochloride profile page.
Summary for CIMETIDINE
| US Patents: | 0 |
| Applicants: | 28 |
| NDAs: | 49 |
| Finished Product Suppliers / Packagers: | 17 |
| Raw Ingredient (Bulk) Api Vendors: | 177 |
| Clinical Trials: | 62 |
| Patent Applications: | 5,745 |
| Formulation / Manufacturing: | see details |
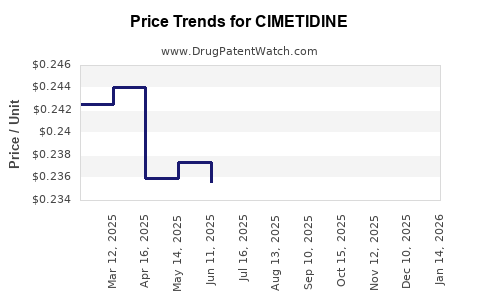
| Drug Prices: | Drug price information for CIMETIDINE |
| DailyMed Link: | CIMETIDINE at DailyMed |
Recent Clinical Trials for CIMETIDINE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Assiut University | Early Phase 1 |
| Chia Tai Tianqing Pharmaceutical Group Co., Ltd. | Phase 2 |
| Beijing Biostar Pharmaceuticals Co., Ltd. | Phase 2 |
Pharmacology for CIMETIDINE
| Drug Class | Histamine-2 Receptor Antagonist |
| Mechanism of Action | Histamine H2 Receptor Antagonists |
Medical Subject Heading (MeSH) Categories for CIMETIDINE
Anatomical Therapeutic Chemical (ATC) Classes for CIMETIDINE
US Patents and Regulatory Information for CIMETIDINE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Teva Parenteral | CIMETIDINE HYDROCHLORIDE | cimetidine hydrochloride | INJECTABLE;INJECTION | 074252-001 | Nov 26, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Pliva | CIMETIDINE | cimetidine | TABLET;ORAL | 074566-001 | Feb 27, 1997 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mylan | CIMETIDINE | cimetidine | TABLET;ORAL | 074246-003 | May 17, 1994 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Hospira | CIMETIDINE HYDROCHLORIDE IN SODIUM CHLORIDE 0.9% IN PLASTIC CONTAINER | cimetidine hydrochloride | INJECTABLE;INJECTION | 074468-006 | Dec 29, 1994 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva | CIMETIDINE | cimetidine | TABLET;ORAL | 074365-001 | Feb 28, 1995 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Watson Labs Inc | CIMETIDINE | cimetidine | TABLET;ORAL | 074349-001 | Aug 30, 1996 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


