LACTULOSE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Lactulose, and when can generic versions of Lactulose launch?
Lactulose is a drug marketed by Cumberland Pharms, Ani Pharms, Apozeal Pharms, Aurobindo Pharma Ltd, Chartwell Rx, Fresenius Kabi, Hikma, Lannett Co Inc, Morton Grove, Paco, Pharm Assoc, Vistapharm, Xttrium Labs Inc, Bajaj, Pai Holdings Pharm, Roxane, and Solvay. and is included in twenty-two NDAs.
The generic ingredient in LACTULOSE is lactulose. There are twenty drug master file entries for this compound. Nineteen suppliers are listed for this compound. Additional details are available on the lactulose profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Lactulose
A generic version of LACTULOSE was approved as lactulose by HIKMA on July 3rd, 1995.
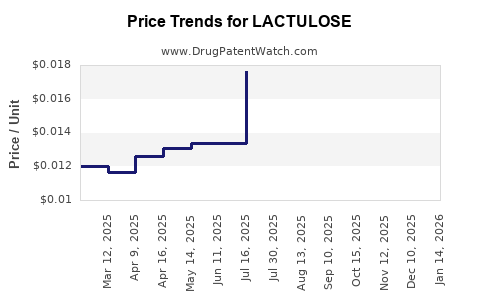
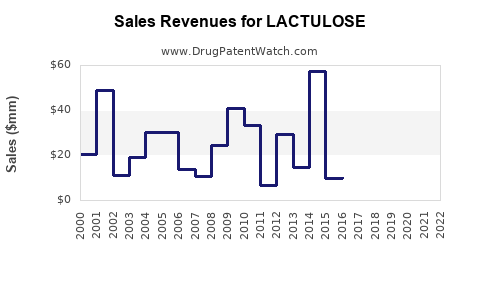
Summary for LACTULOSE
Recent Clinical Trials for LACTULOSE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Michigan | Phase 3 |
| Patient-Centered Outcomes Research Institute | Phase 3 |
| Khyber Medical College, Peshawar | Phase 4 |
Pharmacology for LACTULOSE
| Drug Class | Osmotic Laxative |
| Mechanism of Action | Acidifying Activity Osmotic Activity |
| Physiological Effect | Stimulation Large Intestine Fluid/Electrolyte Secretion |
Medical Subject Heading (MeSH) Categories for LACTULOSE
Anatomical Therapeutic Chemical (ATC) Classes for LACTULOSE
US Patents and Regulatory Information for LACTULOSE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cumberland Pharms | LACTULOSE | lactulose | FOR SOLUTION;ORAL | 074712-001 | Dec 10, 1997 | RX | No | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bajaj | LACTULOSE | lactulose | SOLUTION;ORAL, RECTAL | 076645-001 | Jul 28, 2003 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Fresenius Kabi | LACTULOSE | lactulose | SOLUTION;ORAL | 090503-001 | Jan 25, 2012 | AA | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Apozeal Pharms | LACTULOSE | lactulose | SOLUTION;ORAL | 207786-001 | Jun 11, 2018 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Paco | LACTULOSE | lactulose | SOLUTION;ORAL | 073160-001 | Aug 25, 1992 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Paco | LACTULOSE | lactulose | SOLUTION;ORAL, RECTAL | 072029-001 | Aug 25, 1992 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


