LETROZOLE Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Letrozole, and what generic alternatives are available?
Letrozole is a drug marketed by Accord Hlthcare, Actavis Totowa, Apotex Inc, Beijing Yiling, Carnegie, Chartwell Rx, Eugia Pharma, Fresenius Kabi Usa, Hikma, Hikma Pharms, Impax Labs, Indicus Pharma, Lannett Co Inc, Mylan, Natco Pharma Ltd, Strides Pharma, Sun Pharm Inds Ltd, Synthon Pharms, and Teva Pharms. and is included in twenty NDAs.
The generic ingredient in LETROZOLE is letrozole. There are twenty-four drug master file entries for this compound. Fifteen suppliers are listed for this compound. Additional details are available on the letrozole profile page.
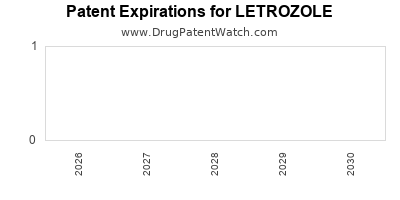
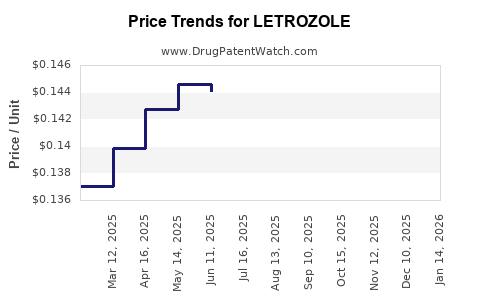
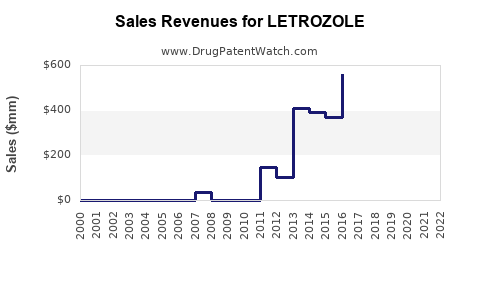
DrugPatentWatch® Litigation and Generic Entry Outlook for Letrozole
A generic version of LETROZOLE was approved as letrozole by ACCORD HLTHCARE on June 3rd, 2011.
Summary for LETROZOLE
Recent Clinical Trials for LETROZOLE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Kom Op Tegen Kanker | Phase 2 |
| Universitaire Ziekenhuizen KU Leuven | Phase 2 |
| University of Miami | Phase 2 |
Pharmacology for LETROZOLE
| Drug Class | Aromatase Inhibitor |
| Mechanism of Action | Aromatase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for LETROZOLE
US Patents and Regulatory Information for LETROZOLE
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Accord Hlthcare | LETROZOLE | letrozole | TABLET;ORAL | 090934-001 | Jun 3, 2011 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Sun Pharm Inds Ltd | LETROZOLE | letrozole | TABLET;ORAL | 091466-001 | Jun 3, 2011 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Eugia Pharma | LETROZOLE | letrozole | TABLET;ORAL | 211717-001 | Jan 11, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Beijing Yiling | LETROZOLE | letrozole | TABLET;ORAL | 205869-001 | Nov 14, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Indicus Pharma | LETROZOLE | letrozole | TABLET;ORAL | 201804-001 | Jun 3, 2011 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Teva Pharms | LETROZOLE | letrozole | TABLET;ORAL | 090289-001 | Jun 3, 2011 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


