OMEGA-3-ACID ETHYL ESTERS Drug Patent Profile
✉ Email this page to a colleague
When do Omega-3-acid Ethyl Esters patents expire, and when can generic versions of Omega-3-acid Ethyl Esters launch?
Omega-3-acid Ethyl Esters is a drug marketed by Amneal Pharms, Apotex, Ascent Pharms Inc, Chartwell, Cspc-nbp Pharm, GLW, Mankind Pharma, Puracap Pharm Llc, Slayback Pharma Llc, Sofgen Pharms, Strides Pharma, Sun Pharm, and Zydus Lifesciences. and is included in fifteen NDAs.
The generic ingredient in OMEGA-3-ACID ETHYL ESTERS is omega-3-acid ethyl esters. There are thirteen drug master file entries for this compound. Twenty-one suppliers are listed for this compound. Additional details are available on the omega-3-acid ethyl esters profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Omega-3-acid Ethyl Esters
A generic version of OMEGA-3-ACID ETHYL ESTERS was approved as omega-3-acid ethyl esters by GLW on April 7th, 2014.
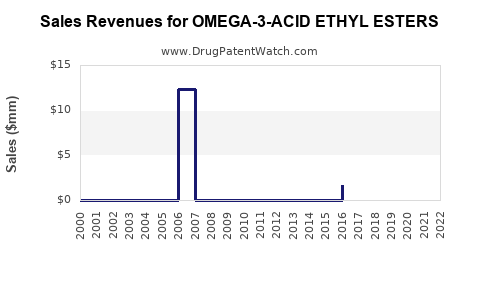
Summary for OMEGA-3-ACID ETHYL ESTERS


See drug prices for OMEGA-3-ACID ETHYL ESTERS
Recent Clinical Trials for OMEGA-3-ACID ETHYL ESTERS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Sohag University | Phase 2/Phase 3 |
| Jinnah Postgraduate Medical Centre | N/A |
| Liaquat National Hospital & Medical College | N/A |
Pharmacology for OMEGA-3-ACID ETHYL ESTERS
| Drug Class | Omega-3 Fatty Acid |
Paragraph IV (Patent) Challenges for OMEGA-3-ACID ETHYL ESTERS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| LOVAZA | Capsules | omega-3-acid ethyl esters | 1 g | 021654 | 3 | 2008-11-10 |
US Patents and Regulatory Information for OMEGA-3-ACID ETHYL ESTERS
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amneal Pharms | OMEGA-3-ACID ETHYL ESTERS | omega-3-acid ethyl esters | CAPSULE;ORAL | 204940-001 | Nov 27, 2015 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Glw | OMEGA-3-ACID ETHYL ESTERS | omega-3-acid ethyl esters | CAPSULE;ORAL | 212504-001 | Aug 19, 2020 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Chartwell | OMEGA-3-ACID ETHYL ESTERS | omega-3-acid ethyl esters | CAPSULE;ORAL | 206455-001 | Aug 7, 2019 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |


