TRIUMEQ Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Triumeq, and when can generic versions of Triumeq launch?
Triumeq is a drug marketed by Viiv Hlthcare and is included in two NDAs. There are two patents protecting this drug and one Paragraph IV challenge.
This drug has one hundred and fifty-five patent family members in thirty-five countries.
The generic ingredient in TRIUMEQ is abacavir sulfate; dolutegravir sodium; lamivudine. There are twelve drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the abacavir sulfate; dolutegravir sodium; lamivudine profile page.
DrugPatentWatch® Generic Entry Outlook for Triumeq
Triumeq was eligible for patent challenges on August 12, 2017.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be June 8, 2030. This may change due to patent challenges or generic licensing.
There have been twenty-two patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
Indicators of Generic Entry
Summary for TRIUMEQ
| International Patents: | 155 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 2 |
| Clinical Trials: | 19 |
| Patent Applications: | 117 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for TRIUMEQ |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for TRIUMEQ |
| What excipients (inactive ingredients) are in TRIUMEQ? | TRIUMEQ excipients list |
| DailyMed Link: | TRIUMEQ at DailyMed |
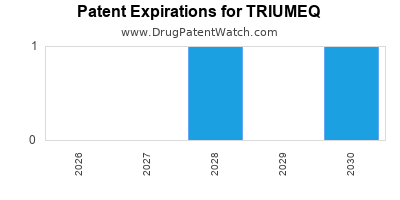
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for TRIUMEQ
Generic Entry Date for TRIUMEQ*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for TRIUMEQ
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| King's College London | Phase 3 |
| Macquarie University, Australia | Phase 3 |
| Stichting TRICALS Foundation | Phase 3 |
Pharmacology for TRIUMEQ
Anatomical Therapeutic Chemical (ATC) Classes for TRIUMEQ
Paragraph IV (Patent) Challenges for TRIUMEQ
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| TRIUMEQ | Tablets | abacavir sulfate; dolutegravir sodium; lamivudine | 600 mg/50 mg/ 300 mg | 205551 | 5 | 2017-08-14 |
US Patents and Regulatory Information for TRIUMEQ
TRIUMEQ is protected by two US patents and two FDA Regulatory Exclusivities.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of TRIUMEQ is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting TRIUMEQ
Substituted 5-hydroxy-3,4,6,9,9a, 10-hexanhydro-2h-1-oxa04a,8a-diaza-anthracene-6,10-dioness
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Synthesis of carbamoylpyridone HIV integrase inhibitors and intermediates
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting TRIUMEQ
INFORMATION ADDED TO THE LABELING REGARDING THE RESULT OF STUDY 205860
Exclusivity Expiration: ⤷ Try a Trial
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Viiv Hlthcare | TRIUMEQ PD | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET, FOR SUSPENSION;ORAL | 215413-001 | Mar 30, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | TRIUMEQ PD | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET, FOR SUSPENSION;ORAL | 215413-001 | Mar 30, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for TRIUMEQ
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| Viiv Hlthcare | TRIUMEQ | abacavir sulfate; dolutegravir sodium; lamivudine | TABLET;ORAL | 205551-001 | Aug 22, 2014 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for TRIUMEQ
When does loss-of-exclusivity occur for TRIUMEQ?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Australia
Patent: 09325128
Estimated Expiration: ⤷ Try a Trial
Patent: 14277831
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0923217
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 44019
Estimated Expiration: ⤷ Try a Trial
Patent: 55957
Estimated Expiration: ⤷ Try a Trial
China
Patent: 2245182
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 76080
Estimated Expiration: ⤷ Try a Trial
Patent: 10603
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 43626
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 86478
Estimated Expiration: ⤷ Try a Trial
Patent: 48595
Estimated Expiration: ⤷ Try a Trial
Patent: 30891
Estimated Expiration: ⤷ Try a Trial
Patent: 12131791
Estimated Expiration: ⤷ Try a Trial
Patent: 12511573
Estimated Expiration: ⤷ Try a Trial
Patent: 16041727
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 1942
Estimated Expiration: ⤷ Try a Trial
Patent: 11006241
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 27451
Estimated Expiration: ⤷ Try a Trial
Patent: 38923
Estimated Expiration: ⤷ Try a Trial
Patent: 11121785
Estimated Expiration: ⤷ Try a Trial
Patent: 13153004
Estimated Expiration: ⤷ Try a Trial
Singapore
Patent: 1308
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1733625
Estimated Expiration: ⤷ Try a Trial
Patent: 1847887
Estimated Expiration: ⤷ Try a Trial
Patent: 110094336
Estimated Expiration: ⤷ Try a Trial
Patent: 170038116
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 41765
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 83947
Estimated Expiration: ⤷ Try a Trial
Patent: 1030010
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering TRIUMEQ around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Canada | 2289654 | ⤷ Try a Trial | |
| Japan | WO2007049675 | HIVインテグラーゼ阻害活性を有する多環性カルバモイルピリドン誘導体 | ⤷ Try a Trial |
| Viet Nam | 34404 | Polycyclic carbamoylpyridone derivative having HIV integrase inhibitory activity | ⤷ Try a Trial |
| Argentina | 012702 | COMBINACION DE (-)-(1S,4R)-4-[2-AMINO-6-(CICLOPROPILAMINO)-9H-PURIN-9-IL]- 2-CICLOPENTENO-1-METANOL Y UN INHIBIDOR DEE TRANSCRIPTASA REVERSA NO NUCLEOSIDOUNA FORMULACION FARMACEUTICA QUE COMPRENDE LA COMBINACION, SU USO MEDICO EN TERAPIA PARA TRATAR LA INFECCION DE VIH, Y UN EMPAQUE PARA EL PACIENTE | ⤷ Try a Trial |
| Mexico | 9707316 | COMBINACIONES SINERGISTICAS DE ZIDOVUDINA, 1592U89 Y 3TC O FTC. (SYNERGISTIC COMBINATIONS OF ZIDOVUDINE, 1592U89 AND 3TC OR FTC.) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for TRIUMEQ
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2465580 | C20210013 00397 | Estonia | ⤷ Try a Trial | PRODUCT NAME: KABOTEGRAVIIR;REG NO/DATE: EU/1/120/1481; 21.12.2020 |
| 0817637 | 300195 | Netherlands | ⤷ Try a Trial | 300195, 20160328, EXPIRES: 20191216 |
| 0817637 | 288 | Finland | ⤷ Try a Trial | |
| 2465580 | LUC00210 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: CABOTEGRAVIR; AUTHORISATION NUMBER AND DATE: EU/1/20/1481 20201221 |
| 0434450 | SPC/GB99/032 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ABACAVIR, OPTIONALLY IN THE FORM OF A PHARMACEUTICALLY ACCEPTABLE SALT OR DERIVATIVE INCLUDING ABACAVIR SULPHATE; REGISTERED: CH CH 55048 001 19990628; CH CH 55049 002 19990628; UK EU/1/99/112/001 19990708; UK EU/1/99/112/002 19990708 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


