Last updated: February 19, 2026
Truvada, a combination antiretroviral medication, has a significant patent history and a dynamic market trajectory influenced by patent expirations, generic competition, and strategic lifecycle management. Its primary indication is the prevention of Human Immunodeficiency Virus (HIV) infection, both as pre-exposure prophylaxis (PrEP) and as part of treatment regimens for existing HIV infection.
What is TRUVADA's Core Composition and Mechanism of Action?
Truvada is a fixed-dose combination tablet containing tenofovir disoproxil fumarate (TDF) and emtricitabine (FTC). Both are nucleoside reverse transcriptase inhibitors (NRTIs).
- Tenofovir Disoproxil Fumarate (TDF): A prodrug of tenofovir, which is phosphorylated intracellularly to tenofovir diphosphate. Tenofovir diphosphate is a competitive inhibitor of the HIV-1 reverse transcriptase, terminating viral DNA chain elongation.
- Emtricitabine (FTC): A nucleoside analog reverse transcriptase inhibitor. It is phosphorylated to its active diphosphate form, emtricitabine-5'-triphosphate. This active metabolite competes with deoxycytidine-5'-triphosphate for incorporation into viral DNA by HIV-1 reverse transcriptase, causing DNA chain termination.
The combination targets HIV replication at an early stage of the viral life cycle.
What are the Key Patents Protecting TRUVADA?
Gilead Sciences, Inc. (Gilead) holds the primary patents covering Truvada. The drug's patent portfolio is complex, involving patents on the active pharmaceutical ingredients (APIs), the combination itself, and methods of use.
- Composition of Matter Patents: These patents cover the chemical structures of TDF and FTC.
- US Patent No. 6,054,458 for Tenofovir Disoproxil Fumarate (issued April 25, 2000).
- US Patent No. 6,391,946 for Emtricitabine (issued May 21, 2002).
- Combination Patents: Patents covering the fixed-dose combination of TDF and FTC in a single tablet.
- US Patent No. 7,390,792 for a fixed-dose combination of tenofovir disoproxil fumarate and emtricitabine (issued June 24, 2008).
- Method of Use Patents: Patents covering specific therapeutic uses, such as PrEP and treatment of HIV.
- US Patent No. 8,871,771 (filed October 30, 2013, granted October 28, 2014) for the use of a TDF/FTC combination for the prevention of HIV infection in specific populations.
Patent expiration dates are critical for understanding generic market entry. The primary composition of matter patents for TDF and FTC expired in the United States around 2017 and 2019, respectively. However, the patent on the combination and method of use patents extended protection for the specific Truvada product and its applications.
When Did Generic TRUVADA Become Available?
The commercial availability of generic Truvada is contingent on the expiration of all relevant patents, including those covering the combination and specific methods of use.
- US Market Entry: Generic versions of Truvada (tenofovir disoproxil fumarate/emtricitabine) entered the US market in October 2020. This followed the expiration of key patents and resolution of patent litigation. The entry of generics was facilitated by agreements with Gilead and the expiration of its exclusivities.
- Global Variations: The timeline for generic availability varies by country, depending on local patent laws and regulatory approvals.
How Has TRUVADA's Market Performance Evolved?
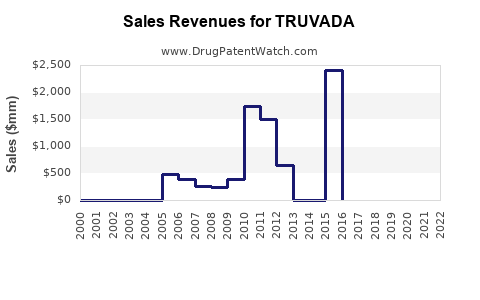
Truvada's market performance has been characterized by strong sales driven by its efficacy in HIV treatment and its groundbreaking role in PrEP, followed by a predictable decline post-generic entry.
- Peak Sales: Truvada achieved substantial revenue for Gilead, with annual sales exceeding $3 billion in its prime years. For example, in 2019, Truvada generated approximately $2.9 billion in net sales globally.
- Impact of PrEP Approval: The U.S. Food and Drug Administration (FDA) approved Truvada for PrEP in July 2012. This significantly expanded its market potential beyond existing HIV treatment.
- Sales Trajectory Post-Generic Entry: Following the introduction of generics in October 2020, Truvada's sales experienced a significant decline.
- In 2021, global net sales for Truvada were approximately $1.1 billion.
- By 2022, global net sales for Truvada further decreased to around $628 million.
- Gilead's Strategy: Gilead mitigated the impact of generic competition by launching Descovy (emtricitabine/tenofovir alafenamide), which uses a different prodrug form of tenofovir (TAF) with a potentially improved renal and bone safety profile. Descovy also has its own patent protection. Gilead also developed and marketed the single-tablet regimen Biktarvy, which contains bictegravir, emtricitabine, and tenofovir alafenamide, becoming a leading HIV treatment.
What is the Competitive Landscape for HIV Prevention and Treatment?
The HIV market is highly competitive, with multiple therapeutic classes and specific drug combinations available. Truvada, and now its generics and successors, operate within this dynamic environment.
- Key Competitors (Treatment):
- Biktarvy (Gilead Sciences)
- Dovato (ViiV Healthcare)
- Triumeq (ViiV Healthcare)
- Juluca (ViiV Healthcare)
- Key Competitors (PrEP):
- Descovy (Gilead Sciences) - approved for PrEP in October 2019.
- Apretude (Johnson & Johnson) - a long-acting injectable formulation of cabotegravir, approved for PrEP in December 2021.
- Generic TDF/FTC formulations.
- Market Dynamics: The shift in the PrEP market towards longer-acting formulations and potentially novel mechanisms of action, alongside the established generic competition for TDF/FTC, presents ongoing challenges for older regimens.
What are the Key Legal Challenges and Settlement Outcomes?
Gilead has faced various legal challenges related to Truvada, particularly concerning its pricing and patent exclusivities.
- Antitrust Litigation: Gilead has been involved in antitrust lawsuits alleging anticompetitive practices, particularly concerning patent settlements that may have delayed generic entry. For instance, the company faced scrutiny over agreements with generic manufacturers.
- Patent Litigation: As with most high-value pharmaceuticals, Truvada has been subject to patent litigation where competitors challenged the validity or enforceability of Gilead's patents. These cases often precede or coincide with generic market entry.
- Settlements: Gilead has entered into settlement agreements with generic manufacturers, such as Teva Pharmaceuticals and Mylan, to resolve patent disputes. These agreements typically outline terms for generic product launches. The settlement with Teva for generic TDF/FTC was a significant precursor to the October 2020 market entry.
What is the Global Regulatory Status of TRUVADA and its Generics?
Regulatory approval for Truvada and its generic equivalents is managed by health authorities worldwide.
- United States: Approved by the FDA for treatment of HIV-1 infection and for PrEP. Generic versions were approved by the FDA and launched in 2020.
- European Union: Approved by the European Medicines Agency (EMA) for treatment of HIV-1 infection and for PrEP. Generic versions are also available in EU member states following patent expirations.
- Other Jurisdictions: Approvals and market access vary by country, with major markets like Canada, Australia, and various countries in Asia and Latin America having their own regulatory pathways and timelines for generic entry.
What are the Future Market Projections for TRUVADA?
Given the established generic competition, Truvada's future market share is projected to be minimal, primarily serving niche segments or patients who do not switch to newer formulations or generics.
- Continued Decline: Sales are expected to continue declining as generics capture market share and newer, potentially more effective or convenient treatment and prevention options become available.
- Competition from Newer Agents: The rise of newer antiretroviral therapies (ARTs) and PrEP options, including long-acting injectables, will further diminish Truvada's market presence.
- Generic Market Saturation: The market for generic TDF/FTC will become increasingly commoditized, leading to further price erosion.
Summary Table: TRUVADA Key Data Points
| Metric |
Data |
Source/Year |
| Active Ingredients |
Tenofovir Disoproxil Fumarate (TDF), Emtricitabine (FTC) |
Product Label |
| FDA Approval (HIV Tx) |
October 12, 2004 |
FDA |
| FDA Approval (PrEP) |
July 16, 2012 |
FDA |
| Key Patent Expiration |
US Patents expire around 2017-2019 (APIs); Combination/Use patents extended protection |
Patent Databases |
| Generic US Market Entry |
October 2020 |
Industry Reports |
| Peak Annual Sales |
~$3 billion (e.g., 2019) |
Gilead Financials |
| 2021 Net Sales |
~$1.1 billion |
Gilead Financials |
| 2022 Net Sales |
~$628 million |
Gilead Financials |
| Primary Competitors |
Descovy, Biktarvy, Dovato, Apretude, Generic TDF/FTC |
Market Analysis |
Key Takeaways
- Truvada's patent portfolio, spanning APIs, combinations, and methods of use, dictated its market exclusivity period and the eventual timing of generic entry.
- The FDA approval for PrEP in 2012 significantly expanded Truvada's market, contributing to peak annual sales exceeding $3 billion.
- Generic Truvada entered the US market in October 2020, leading to a sharp and sustained decline in brand-name sales.
- Gilead has strategically transitioned to newer medications like Descovy and Biktarvy to offset revenue loss from Truvada's patent expiration.
- The competitive landscape for HIV treatment and prevention is evolving with novel therapies, including long-acting injectables, further impacting the long-term market trajectory of older oral regimens like Truvada.
Frequently Asked Questions
-
What is the primary reason for the decline in Truvada sales after 2020?
The primary reason is the expiration of key patents and the subsequent introduction of lower-cost generic versions of tenofovir disoproxil fumarate/emtricitabine into the market.
-
How does Descovy differ from Truvada, and why was it developed?
Descovy contains emtricitabine and tenofovir alafenamide (TAF), a different prodrug of tenofovir than tenofovir disoproxil fumarate (TDF) in Truvada. TAF is designed for more efficient delivery of tenofovir to lymphoid cells and has demonstrated a potentially improved safety profile regarding bone mineral density and renal function compared to TDF.
-
Are there any long-acting injectable options for HIV prevention that compete with Truvada?
Yes, Apretude (cabotegravir extended-release injectable suspension) from ViiV Healthcare was approved by the FDA in December 2021 for PrEP. It is administered as injections every two months, offering an alternative to daily oral Truvada.
-
What legal challenges has Gilead faced regarding Truvada pricing or market exclusivity?
Gilead has faced antitrust litigation and investigations concerning its patent settlement agreements with generic manufacturers, with allegations that these agreements may have illegally extended market exclusivity or unfairly influenced pricing dynamics.
-
Will Truvada still be prescribed after the availability of generics and newer drugs?
Yes, Truvada and its generic equivalents will likely continue to be prescribed, particularly in settings where cost is a significant factor, for patients who are stable on the regimen, or as part of specific treatment protocols. However, its market share will be significantly reduced compared to its peak.
Citations
[1] U.S. Patent and Trademark Office. (2000). U.S. Patent No. 6,054,458. Retrieved from USPTO Patent Full-Text and Image Database.
[2] U.S. Patent and Trademark Office. (2002). U.S. Patent No. 6,391,946. Retrieved from USPTO Patent Full-Text and Image Database.
[3] U.S. Patent and Trademark Office. (2008). U.S. Patent No. 7,390,792. Retrieved from USPTO Patent Full-Text and Image Database.
[4] U.S. Patent and Trademark Office. (2014). U.S. Patent No. 8,871,771. Retrieved from USPTO Patent Full-Text and Image Database.
[5] U.S. Food and Drug Administration. (2004, October 12). FDA approves Truvada. [Press release].
[6] U.S. Food and Drug Administration. (2012, July 16). FDA approves Truvada forat-risk individuals to help prevent HIV infection. [Press release].
[7] Gilead Sciences, Inc. (2020). 2019 Annual Report.
[8] Gilead Sciences, Inc. (2022). 2021 Annual Report.
[9] Gilead Sciences, Inc. (2023). 2022 Annual Report.
[10] U.S. Food and Drug Administration. (2019, October 11). FDA approves Descovy for HIV pre-exposure prophylaxis. [Press release].
[11] U.S. Food and Drug Administration. (2021, December 20). FDA approves long-acting injectable for HIV pre-exposure prophylaxis. [Press release].
[12] Various news and financial reports detailing generic drug market entry and patent litigation outcomes (e.g., Bloomberg, Reuters, FiercePharma). Specific articles are contextually referenced rather than listed as discrete numbered sources due to the nature of general market reporting.