YONDELIS Drug Patent Profile
✉ Email this page to a colleague
When do Yondelis patents expire, and when can generic versions of Yondelis launch?
Yondelis is a drug marketed by Janssen Prods and is included in one NDA. There is one patent protecting this drug and one Paragraph IV challenge.
This drug has forty-four patent family members in thirty-six countries.
The generic ingredient in YONDELIS is trabectedin. There are four drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the trabectedin profile page.
DrugPatentWatch® Generic Entry Outlook for Yondelis
Yondelis was eligible for patent challenges on October 23, 2019.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be July 7, 2028. This may change due to patent challenges or generic licensing.
There have been three patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There is one tentative approval for the generic drug (trabectedin), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
Summary for YONDELIS
| International Patents: | 44 |
| US Patents: | 1 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 43 |
| Clinical Trials: | 51 |
| Patent Applications: | 2,970 |
| Drug Prices: | Drug price information for YONDELIS |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for YONDELIS |
| What excipients (inactive ingredients) are in YONDELIS? | YONDELIS excipients list |
| DailyMed Link: | YONDELIS at DailyMed |
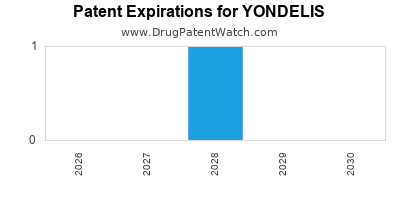

DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for YONDELIS
Generic Entry Date for YONDELIS*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INTRAVENOUS |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for YONDELIS
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Westfälische Wilhelms-Universität Münster | Phase 3 |
| Anturec Pharmaceuticals GmbH | Phase 3 |
| Rain Therapeutics Inc. | Phase 3 |
Pharmacology for YONDELIS
| Drug Class | Alkylating Drug |
| Mechanism of Action | Alkylating Activity |
Anatomical Therapeutic Chemical (ATC) Classes for YONDELIS
Paragraph IV (Patent) Challenges for YONDELIS
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| YONDELIS | Powder for Injection | trabectedin | 1 mg/vial | 207953 | 2 | 2020-04-23 |
US Patents and Regulatory Information for YONDELIS
YONDELIS is protected by one US patents.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of YONDELIS is ⤷ Try a Trial.
This potential generic entry date is based on patent ⤷ Try a Trial.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
Patents protecting YONDELIS
Pharmaceutical formulations of ecteinascidin compounds
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Janssen Prods | YONDELIS | trabectedin | POWDER;INTRAVENOUS | 207953-001 | Oct 23, 2015 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for YONDELIS
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Pharma Mar S.A. | Yondelis | trabectedin | EMEA/H/C/000773 Yondelis is indicated for the treatment of patients with advanced soft-tissue sarcoma, after failure of anthracyclines and ifosfamide, or who are unsuited to receive these agents. Efficacy data are based mainly on liposarcoma and leiomyosarcoma patients.Yondelis in combination with pegylated liposomal doxorubicin (PLD) is indicated for the treatment of patients with relapsed platinum-sensitive ovarian cancer. |
Authorised | no | no | no | 2007-09-17 | |
| Pharma Mar S.A. | Yondelis | trabectedin | EMEA/H/C/000464 Yondelis is indicated for the treatment of patients with advanced soft tissue sarcoma, having failed antracyclines and ifosfamide, or having failed ifosfamide and unsuitable to receive antracyclines/ifosfamide. |
Refused | no | no | no | 2004-09-07 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for YONDELIS
When does loss-of-exclusivity occur for YONDELIS?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
Argentina
Patent: 1653
Estimated Expiration: ⤷ Try a Trial
Patent: 0986
Estimated Expiration: ⤷ Try a Trial
Australia
Patent: 05227421
Estimated Expiration: ⤷ Try a Trial
Austria
Patent: 68461
Estimated Expiration: ⤷ Try a Trial
Brazil
Patent: 0517238
Estimated Expiration: ⤷ Try a Trial
Canada
Patent: 20979
Estimated Expiration: ⤷ Try a Trial
China
Patent: 23794
Estimated Expiration: ⤷ Try a Trial
Patent: 2018714
Estimated Expiration: ⤷ Try a Trial
Croatia
Patent: 0070374
Estimated Expiration: ⤷ Try a Trial
Cyprus
Patent: 07773
Estimated Expiration: ⤷ Try a Trial
Denmark
Patent: 58848
Estimated Expiration: ⤷ Try a Trial
El Salvador
Patent: 06002283
Estimated Expiration: ⤷ Try a Trial
European Patent Office
Patent: 58848
Estimated Expiration: ⤷ Try a Trial
Germany
Patent: 2005001833
Estimated Expiration: ⤷ Try a Trial
Guatemala
Patent: 0500314
Estimated Expiration: ⤷ Try a Trial
Honduras
Patent: 05029978
Estimated Expiration: ⤷ Try a Trial
Hong Kong
Patent: 91724
Estimated Expiration: ⤷ Try a Trial
Israel
Patent: 2776
Estimated Expiration: ⤷ Try a Trial
Japan
Patent: 68163
Estimated Expiration: ⤷ Try a Trial
Patent: 06124393
Estimated Expiration: ⤷ Try a Trial
Jordan
Patent: 64
Estimated Expiration: ⤷ Try a Trial
Malaysia
Patent: 9049
Estimated Expiration: ⤷ Try a Trial
Mexico
Patent: 05011650
Estimated Expiration: ⤷ Try a Trial
New Zealand
Patent: 4761
Estimated Expiration: ⤷ Try a Trial
Norway
Patent: 8035
Estimated Expiration: ⤷ Try a Trial
Patent: 072713
Estimated Expiration: ⤷ Try a Trial
Panama
Patent: 50801
Estimated Expiration: ⤷ Try a Trial
Peru
Patent: 060925
Estimated Expiration: ⤷ Try a Trial
Patent: 100089
Estimated Expiration: ⤷ Try a Trial
Poland
Patent: 58848
Estimated Expiration: ⤷ Try a Trial
Portugal
Patent: 58848
Estimated Expiration: ⤷ Try a Trial
Russian Federation
Patent: 82647
Estimated Expiration: ⤷ Try a Trial
Patent: 07119724
Estimated Expiration: ⤷ Try a Trial
Serbia
Patent: 510
Estimated Expiration: ⤷ Try a Trial
Slovenia
Patent: 58848
Estimated Expiration: ⤷ Try a Trial
South Africa
Patent: 0703450
Estimated Expiration: ⤷ Try a Trial
South Korea
Patent: 1188691
Estimated Expiration: ⤷ Try a Trial
Patent: 070091121
Estimated Expiration: ⤷ Try a Trial
Spain
Patent: 90844
Estimated Expiration: ⤷ Try a Trial
Taiwan
Patent: 96523
Estimated Expiration: ⤷ Try a Trial
Patent: 0621256
Estimated Expiration: ⤷ Try a Trial
Ukraine
Patent: 032
Estimated Expiration: ⤷ Try a Trial
Uruguay
Patent: 183
Estimated Expiration: ⤷ Try a Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering YONDELIS around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Panama | 8650801 | COMPOSICIONES Y FORMULACIONES DE ECTEINASCIDINA, COMO LA ECTEINASCIDINA 743 | ⤷ Try a Trial |
| Austria | E368461 | ⤷ Try a Trial | |
| Japan | 2006124393 | FORMULATION | ⤷ Try a Trial |
| Serbia | 50510 | FORMULACIJE KOJE SADRŽE EKTEINASCIDIN I DISAHARID | ⤷ Try a Trial |
| Guatemala | 200500314 | FORMULACIONES | ⤷ Try a Trial |
| New Zealand | 554761 | Formulations comprising ecteinascidin and a disaccharide | ⤷ Try a Trial |
| Spain | 2290844 | ⤷ Try a Trial | |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.


