AMICUS THERAP US Company Profile
✉ Email this page to a colleague
What is the competitive landscape for AMICUS THERAP US
AMICUS THERAP US has two approved drugs.
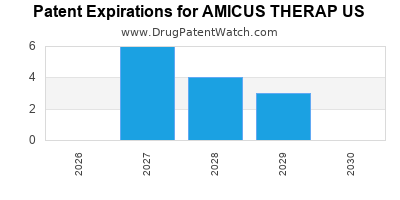
There are seventy-three US patents protecting AMICUS THERAP US drugs.
There are four hundred and thirty-six patent family members on AMICUS THERAP US drugs in forty-five countries and thirty-four supplementary protection certificates in eighteen countries.
Summary for AMICUS THERAP US
| International Patents: | 436 |
| US Patents: | 73 |
| Tradenames: | 2 |
| Ingredients: | 2 |
| NDAs: | 2 |
Drugs and US Patents for AMICUS THERAP US
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 10,525,045 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,234,972 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | OPFOLDA | miglustat | CAPSULE;ORAL | 215211-001 | Sep 28, 2023 | RX | Yes | Yes | 10,961,522 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,622,962 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Amicus Therap Us | OPFOLDA | miglustat | CAPSULE;ORAL | 215211-001 | Sep 28, 2023 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,357,761 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Amicus Therap Us | GALAFOLD | migalastat hydrochloride | CAPSULE;ORAL | 208623-001 | Aug 10, 2018 | RX | Yes | Yes | 11,033,538 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for AMICUS THERAP US Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Australia | 2018220047 | ⤷ Start Trial |
| Nicaragua | 201700039 | ⤷ Start Trial |
| Japan | 6875955 | ⤷ Start Trial |
| South Korea | 20230061563 | ⤷ Start Trial |
| Brazil | 112019025083 | ⤷ Start Trial |
| Taiwan | 201733581 | ⤷ Start Trial |
| Taiwan | 201627499 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for AMICUS THERAP US Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2787345 | CR 2016 00055 | Denmark | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT ELLER ET SALT HERAF, HERUNDER HYDROGENKLORIDSALTET; REG. NO/DATE: EU/1/15/1082 20160531 |
| 3201320 | PA2024509 | Lithuania | ⤷ Start Trial | PRODUCT NAME: CIPAGLIUKOZIDAZE ALFA; REGISTRATION NO/DATE: EU/1/22/1714 20230320 |
| 2787345 | C 2016 042 | Romania | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT SAU O SARE A ACESTUIA, INCLUSIV SAREACLORHIDRAT; NATIONAL AUTHORISATION NUMBER: EU/1/15/1082; DATE OF NATIONAL AUTHORISATION: 20160526; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/15/1082; DATE OF FIRST AUTHORISATION IN EEA: 20160526 |
| 4273241 | 301330 | Netherlands | ⤷ Start Trial | PRODUCT NAME: CIPAGLUCOSIDASE ALFA; REGISTRATION NO/DATE: EU/1/22/1714 20230324 |
| 2787345 | PA2016033,C2787345 | Lithuania | ⤷ Start Trial | PRODUCT NAME: MIGALASTATAS ARBA JO DRUSKA, ISKAITANT IR HIDROCHLORIDO DRUSKA; REGISTRATION NO/DATE: EU/1/15/1082 20160526 |
| 2787345 | 268 5025-2016 | Slovakia | ⤷ Start Trial | PRODUCT NAME: MIGALASTAT VO VSETKYCH FORMACH CHRANENYCH ZAKLADNYM PATENTOM; REGISTRATION NO/DATE: EU/1/15/1082 20160531 |
| 4273241 | 122025000021 | Germany | ⤷ Start Trial | PRODUCT NAME: CIPAGLUCOSIDASE ALFA; REGISTRATION NO/DATE: EU/1/22/1714 20230320 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.