KEY THERAP Company Profile
✉ Email this page to a colleague
What is the competitive landscape for KEY THERAP, and what generic alternatives to KEY THERAP drugs are available?
KEY THERAP has seven approved drugs.
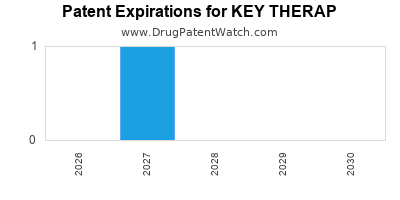
There are two US patents protecting KEY THERAP drugs.
There are one hundred and fifty-five patent family members on KEY THERAP drugs in thirty-seven countries and twenty-four supplementary protection certificates in sixteen countries.
Drugs and US Patents for KEY THERAP
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Key Therap | NALFON | fenoprofen calcium | CAPSULE;ORAL | 017604-004 | Jul 21, 2009 | AB | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | ||||
| Key Therap | CETRAXAL | ciprofloxacin hydrochloride | SOLUTION/DROPS;OTIC | 021918-001 | May 1, 2009 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | |||||
| Key Therap | BUTALBITAL, ACETAMINOPHEN AND CAFFEINE | acetaminophen; butalbital; caffeine | CAPSULE;ORAL | 206615-001 | Aug 4, 2017 | AA | RX | No | No | ⤷ Sign Up | ⤷ Sign Up | ||||
| Key Therap | NALFON | fenoprofen calcium | CAPSULE;ORAL | 017604-002 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Key Therap | ZONTIVITY | vorapaxar sulfate | TABLET;ORAL | 204886-001 | May 8, 2014 | RX | Yes | Yes | 7,304,078 | ⤷ Sign Up | Y | Y | ⤷ Sign Up | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for KEY THERAP
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Key Therap | NALFON | fenoprofen calcium | CAPSULE;ORAL | 017604-003 | Approved Prior to Jan 1, 1982 | 3,600,437 | ⤷ Sign Up |
| Key Therap | ZONTIVITY | vorapaxar sulfate | TABLET;ORAL | 204886-001 | May 8, 2014 | 7,235,567 | ⤷ Sign Up |
| Key Therap | ZONTIVITY | vorapaxar sulfate | TABLET;ORAL | 204886-001 | May 8, 2014 | 7,713,999 | ⤷ Sign Up |
| Key Therap | NALFON | fenoprofen calcium | CAPSULE;ORAL | 017604-002 | Approved Prior to Jan 1, 1982 | 3,600,437 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for KEY THERAP Drugs
Supplementary Protection Certificates for KEY THERAP Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1429780 | SPC/GB12/058 | United Kingdom | ⤷ Sign Up | PRODUCT NAME: A COMBINATION OF CIPROFLOXACIN AND DEXAMETHASONE, PREFERABLY CIPROFLOXACIN HYDROCHLORIDE AND DEXAMETHASONE; REGISTERED: DK DE/11/3337/001/DC 20120808; UK PL000649/0381-0001 20121003 |
| 1495018 | 122015000053 | Germany | ⤷ Sign Up | PRODUCT NAME: VORAPAXAR ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ ODER SOLVAT DAVON; REGISTRATION NO/DATE: EU/1/14/976/001-006 20150119 |
| 1495018 | 34/2015 | Austria | ⤷ Sign Up | PRODUCT NAME: VORAPAXAR, ODER EIN PHARMAZEUTISCH ANNEHMBARES SALZ ODER SOLVAT DAVON; REGISTRATION NO/DATE: EU/1/14/976 20150119 |
| 1495018 | PA2015027 | Lithuania | ⤷ Sign Up | PRODUCT NAME: VORAPAXARUM; REGISTRATION NO/DATE: EU/1/14/976/001 - EU/1/14/976/006 20150119 |
| 1495018 | 300746 | Netherlands | ⤷ Sign Up | PRODUCT NAME: VORAPAXAR, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF SOLVAAT, IN HET BIJZONDER VORAPAXARSULFAAT; REGISTRATION NO/DATE: EU/1/14/976/001-006 20150121 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.