LUPIN LTD Company Profile
✉ Email this page to a colleague
What is the competitive landscape for LUPIN LTD, and what generic alternatives to LUPIN LTD drugs are available?
LUPIN LTD has one hundred and sixty-eight approved drugs.
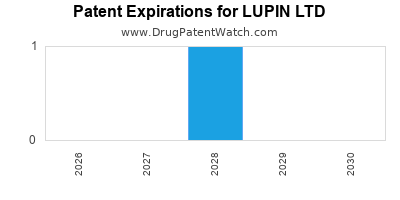
There is one US patent protecting LUPIN LTD drugs. There are twenty-one tentative approvals on LUPIN LTD drugs.
There are two patent family members on LUPIN LTD drugs in two countries and six hundred and ten supplementary protection certificates in seventeen countries.
Summary for LUPIN LTD
| International Patents: | 2 |
| US Patents: | 1 |
| Tradenames: | 145 |
| Ingredients: | 128 |
| NDAs: | 168 |
| Drug Master File Entries: | 194 |
| Patent Litigation for LUPIN LTD: | See patent lawsuits for LUPIN LTD |
| PTAB Cases with LUPIN LTD as petitioner: | See PTAB cases with LUPIN LTD as petitioner |
Drugs and US Patents for LUPIN LTD
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Lupin Ltd | PITAVASTATIN CALCIUM | pitavastatin calcium | TABLET;ORAL | 206029-001 | Nov 20, 2023 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Lupin Ltd | IMIPRAMINE PAMOATE | imipramine pamoate | CAPSULE;ORAL | 090444-004 | Apr 16, 2010 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Lupin Ltd | PAROXETINE HYDROCHLORIDE | paroxetine hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 204134-001 | Jan 20, 2017 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Lupin Ltd | PERINDOPRIL ERBUMINE | perindopril erbumine | TABLET;ORAL | 078263-003 | Jan 27, 2010 | DISCN | No | No | ⤷ Subscribe | ⤷ Subscribe | |||||
| Lupin Ltd | METRONIDAZOLE | metronidazole | TABLET;ORAL | 209096-002 | Sep 12, 2017 | AB | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| Lupin Ltd | DECITABINE | decitabine | INJECTABLE;INTRAVENOUS | 210756-001 | Nov 9, 2018 | AP | RX | No | No | ⤷ Subscribe | ⤷ Subscribe | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Paragraph IV (Patent) Challenges for LUPIN LTD drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Delayed-release Tablets | 20 mg | ➤ Subscribe | 2015-06-03 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
| ➤ Subscribe | for Oral Suspension | 500 mg/5 mL | ➤ Subscribe | 2014-07-22 |
International Patents for LUPIN LTD Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Germany | 112007000920 | ⤷ Subscribe |
| World Intellectual Property Organization (WIPO) | 2007119249 | ⤷ Subscribe |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for LUPIN LTD Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1663240 | PA2015037 | Lithuania | ⤷ Subscribe | PRODUCT NAME: RILPIVIRINUM + TENOFOVIRUM DISOPROXILUM + EMTRICITABINUM; REGISTRATION NO/DATE: EU/1/11/737/001 2011 11 28, EU/1/11/737/002 20111128 |
| 2207786 | 2023C/551 | Belgium | ⤷ Subscribe | PRODUCT NAME: UNE COMPOSITION COMPRENANT : DE LA CEDAZURIDINE OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE DE CELLE-CI ; ET LA DECITABINE; AUTHORISATION NUMBER AND DATE: EU/1/23/1756 20230918 |
| 1663240 | 2015/054 | Ireland | ⤷ Subscribe | PRODUCT NAME: A COMBINATION OF RILPIVIRINE OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, INCLUDING THE HYDROCHLORIDE SALT, AND TENOFOVIR, IN PARTICULAR TENOFOVIR DISOPROXIL FUMARATE; REGISTRATION NO/DATE: EU/1/11/737/001-002 20111128 |
| 0247633 | C970034 | Netherlands | ⤷ Subscribe | PRODUCT NAME: ATORVASTATINUM,DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF VAN HET INWENDIGE DELTA-LACTON, IN HET BIJZONDER ATORVASTATINUM CALCICUM TRIHYDRICUM; NAT. REGISTRATION NO/DATE: RVG 21081 - RVG 21083 19970421; FIRST REGISTRATION: GB PL 00018/0240 - PL 00018/0242 19961107 |
| 1532149 | PA2012022 | Lithuania | ⤷ Subscribe | PRODUCT NAME: LINAGLIPTINUM + METFORMINI HYDROCHLORIDUM; REGISTRATION NO/DATE: EU/1/12/780/001 - EU/1/12/780/034 20120720 |
| 1663240 | PA2015038 | Lithuania | ⤷ Subscribe | PRODUCT NAME: RILPIVIRINUM + TENOFOVIRUM DISOPROXILUM; REGISTRATION NO/DATE: EU/1/11/737/001, 2011 11 28, EU/1/11/737/002 20111128 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.