Share This Page
Drug Price Trends for FT ITCH RELIEF
✉ Email this page to a colleague
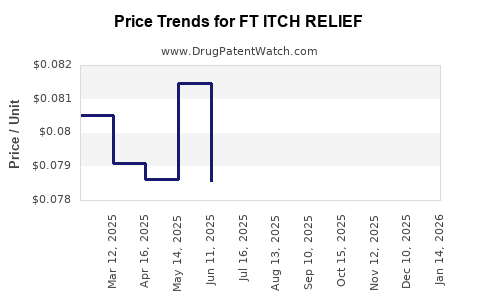
Average Pharmacy Cost for FT ITCH RELIEF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| FT ITCH RELIEF 1% OINTMENT | 70677-1214-01 | 0.07910 | GM | 2025-03-19 |
| FT ITCH RELIEF 1% OINTMENT | 70677-1214-01 | 0.08051 | GM | 2025-02-19 |
| FT ITCH RELIEF 1% OINTMENT | 70677-1214-01 | 0.08058 | GM | 2025-01-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Itch Relief Drugs
Introduction to Itch Relief Drugs
Itch relief drugs, including those for pruritus and anti-itch creams, are a significant segment of the pharmaceutical and healthcare industries. These medications are designed to alleviate the discomfort, irritation, and itching associated with various dermatological and systemic conditions.
Current Market Size and Growth
The market for itch relief drugs is experiencing substantial growth driven by several factors:
U.S. Pruritus Treatment Market
The U.S. pruritus treatment market was valued at approximately $5.1 billion in 2022 and is projected to reach $8.4 billion by 2031, growing at a Compound Annual Growth Rate (CAGR) of 5.6% from 2023 to 2031. This growth is attributed to the increasing availability of innovative therapies such as immunosuppressants, topical corticosteroids, and antihistamines, along with rising awareness of the condition[1].
Global Anti-Itch Cream Market
The global anti-itch cream market was valued at $5.2 billion in 2023 and is expected to reach $10.8 billion by 2030, with a CAGR of 8% during the forecast period from 2024 to 2030. This market includes a wide variety of formulations such as gels, ointments, creams, and lotions designed to relieve itching caused by various dermatological diseases[2].
Global Pruritus Drugs Market
On a broader scale, the global pruritus drugs market was valued at $8.2 billion in 2023 and is predicted to reach $12.5 billion by 2033, growing at a CAGR of 4.4% during the forecast period from 2024 to 2033. This market encompasses pharmaceuticals targeting itch relief, ranging from topical creams to systemic medications[4].
Key Drivers of Market Growth
Several factors are driving the growth of the itch relief drug market:
Increasing Prevalence of Skin Disorders
The rising prevalence of skin disorders such as eczema, psoriasis, and allergic reactions is a significant driver. These conditions are becoming more common, leading to an increased demand for effective itch relief treatments[2].
Innovative Therapies
The development and launch of innovative therapies, including immunosuppressants, topical corticosteroids, and antihistamines, are contributing to market growth. For example, the introduction of new products like Alaway Preservative-Free antihistamine eye drops has expanded treatment options for patients[3].
Rising Awareness
Growing awareness of pruritus and other skin conditions among the general public and healthcare professionals is also driving the market. This increased awareness leads to better diagnosis and treatment rates, further boosting market growth[1].
Market Segments and Key Players
The itch relief drug market is segmented into various product types and therapeutic classes:
Product Types
- Topical creams and ointments
- Gels and lotions
- Systemic medications
- Antihistamine eye drops
Key Players
Leading players in the market include:
- Aveeno
- POLYSPORIN
- CeraVe
- BENADRYL
- VANICREAM
- Bausch + Lomb
- Sanofi's Chattem
- Novartis International AG
- Pfizer Inc.
- Johnson & Johnson[2][3][5].
Price Projections and Market Trends
Price Trends
The prices of itch relief drugs are expected to remain competitive due to the presence of both prescription and over-the-counter (OTC) options. For instance, the Allegra Anti-Itch Cream is priced between $4.99 and $6.99, making it accessible to a wide range of consumers[5].
Market Trends
The market is trending towards more innovative and patient-friendly products. For example, the launch of preservative-free antihistamine eye drops like Alaway Preservative-Free by Bausch + Lomb indicates a shift towards safer and more effective treatments[3].
Regulatory Environment
The regulatory environment plays a crucial role in the development and approval of itch relief drugs. Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) ensure that these products meet stringent safety and efficacy standards before they are approved for market use.
Challenges and Restraints
Despite the growth, the market faces several challenges:
Regulatory Hurdles
The approval process for new drugs can be lengthy and uncertain, posing a significant challenge to market growth. Regulatory decisions can impact the availability and commercial potential of new treatments[5].
Competition
The market is highly competitive, with numerous players offering a range of products. This competition can lead to pricing pressures and make it challenging for new entrants to gain market share[2].
Future Outlook
The future outlook for the itch relief drug market is promising, driven by the increasing demand for effective treatments and the continuous innovation in therapeutic options.
"The availability of innovative therapies such as immunosuppressants, topical corticosteroids, and antihistamines, together with rising awareness of the illness, are expected to support the expansion of the pruritus treatment market in the United States throughout the coming years."[1]
Key Takeaways
- The U.S. pruritus treatment market is expected to reach $8.4 billion by 2031, growing at a CAGR of 5.6%.
- The global anti-itch cream market is projected to reach $10.8 billion by 2030, with a CAGR of 8%.
- The global pruritus drugs market is predicted to reach $12.5 billion by 2033, growing at a CAGR of 4.4%.
- Innovative therapies and rising awareness are key drivers of market growth.
- The market is highly competitive, with several leading players offering a range of products.
FAQs
1. What is the projected size of the U.S. pruritus treatment market by 2031? The U.S. pruritus treatment market is expected to reach $8.4 billion by 2031[1].
2. Which companies are leading the anti-itch cream market? Leading players include Aveeno, POLYSPORIN, CeraVe, BENADRYL, and VANICREAM[2].
3. What is the CAGR of the global anti-itch cream market from 2024 to 2030? The global anti-itch cream market is expected to grow at a CAGR of 8% from 2024 to 2030[2].
4. What are some of the key drivers of the itch relief drug market? Key drivers include the increasing prevalence of skin disorders, innovative therapies, and rising awareness of pruritus and other skin conditions[1][2][4].
5. What is the significance of regulatory approvals in the itch relief drug market? Regulatory approvals are crucial as they ensure the safety and efficacy of new treatments, impacting their availability and commercial potential[5].
Sources
- Transparency Market Research Inc. - U.S. Pruritus Treatment Market to Hit USD 8.4 billion by 2031[1].
- Verified Market Reports - Anti itch Cream Market Size, Share, Growth | Global Industry Trends[2].
- Coherent Market Insights - Viral Conjunctivitis Drugs Market - Price, Size, Share & Growth[3].
- Marketresearch.biz - Pruritus Drugs Market Predicted to Garner USD 12.5 Bn By 2033, At CAGR 4.4%[4].
- Sanofi - New Allegra® Anti-Itch Cream Introduced by Sanofi's Chattem[5].
More… ↓


