Share This Page
Drug Sales Trends for ABILIFY
✉ Email this page to a colleague
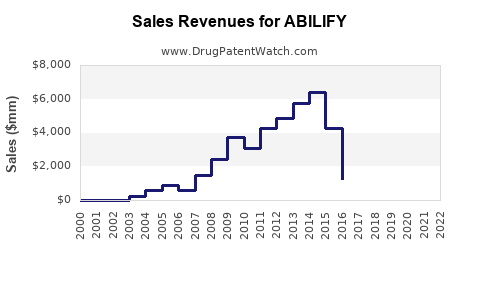
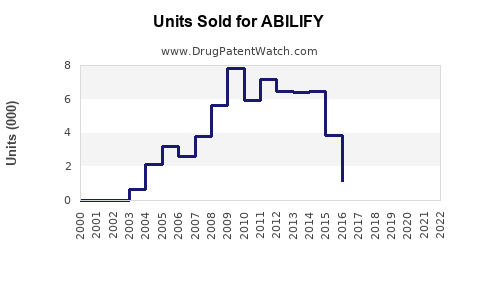
Annual Sales Revenues and Units Sold for ABILIFY
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2021 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2020 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2019 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2018 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2017 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2016 |
| ABILIFY | ⤷ Try for Free | ⤷ Try for Free | 2015 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Abilify (Aripiprazole)
Introduction to Abilify (Aripiprazole)
Abilify, also known as aripiprazole, is a second-generation antipsychotic medication developed by Otsuka Pharmaceutical. It is used to treat various psychiatric conditions, including schizophrenia, bipolar mania, and major depressive disorders. Here’s a comprehensive analysis of the market and sales projections for Abilify.
Current Market Scenario
The global aripiprazole market is expected to experience significant growth in the coming years. As of 2023, the global Aripiprazole Drug market was valued at US$ 121 million and is anticipated to reach US$ 167.1 million by 2030, growing at a CAGR of 5.1% during the forecast period of 2024-2030[2].
Key Drivers of Market Growth
Several factors are driving the growth of the aripiprazole market:
- Increasing Prevalence of Mental Health Disorders: The rise in the prevalence of depression, schizophrenia, and bipolar mania globally is a major driver for the expansion of the aripiprazole market[1].
- Geriatric Population and Healthcare Expenditure: The increasing geriatric population and rising healthcare expenditure are also contributing to market growth[1].
- Research and Development: The rise in research and development activities and the emergence of new markets will provide beneficial opportunities for the aripiprazole market[1].
Market Segmentation
The aripiprazole market is segmented based on several criteria:
- Drug Class: Antipsychotics and antimanic agents[1].
- Application: Major depressive disorders, schizophrenia, bipolar mania, and others[1].
- Demographic: Adult and pediatric patients[2].
- Dosage Form: Tablets, orally disintegrating tablets, oral solution, and injection[1][2].
- End-Users: Clinics, hospitals, and others[1].
- Distribution Channel: Hospital pharmacy, retail pharmacy, and online pharmacy[1].
Major Players
The market is dominated by several key players:
- Otsuka Pharmaceutical
- Teva
- APOTEX
- Barr Laboratories
- Sun Pharmaceutical
- Alembic Pharmaceuticals
- Orchid Pharma
- Amneal
- Aurobindo Pharma
- Lannett
- Solco Healthcare
- Delphis Pharma[2].
Impact of Patent Expiry
A significant event affecting the market is the impending loss of market exclusivity for Abilify Maintena in Europe and the US in October 2024. This will lead to the introduction of generic versions, which is expected to result in a decline in sales for Otsuka. The sales of Abilify Maintena are forecasted to drop from $1.5 billion in 2024 to $459 million by the end of 2029, with a negative CAGR of 21% from 2024 to 2029[4].
New Developments and Strategies
To mitigate the impact of patent expiry, Otsuka and Lundbeck have received FDA approval for Abilify Asimtufii, a longer-acting version of the drug administered every two months. This new formulation has shown comparable safety and efficacy to Abilify Maintena and is expected to help Otsuka maintain its market position despite the introduction of generics[5].
Sales Projections
- Abilify Maintena: Despite the decline due to patent expiry, Abilify Maintena remains a significant player. Its sales were $419 million in 2022, a 20% increase from 2021. However, sales are projected to decline significantly post-patent expiry[5].
- Global Aripiprazole Market: The market is expected to grow from US$ 121 million in 2023 to US$ 167.1 million by 2030, with a CAGR of 5.1% during the forecast period[2].
Regional Market Analysis
The market analysis varies by region:
- United States: The US is one of the most important markets for Otsuka, contributing 49% of the drug’s total historical revenue from 2013 to 2023[4].
- Europe: The loss of market exclusivity in Europe will also impact sales significantly[4].
- Other Regions: The market is also growing in other regions, including Asia-Pacific, driven by increasing healthcare expenditure and rising awareness of mental health disorders[1].
Competitive Landscape
The competitive landscape is set to change with the introduction of generics and new therapies:
- Generics: The introduction of generic versions of aripiprazole will increase competition and reduce prices, making the drug more accessible to a larger patient population[4].
- Emerging Therapies: Other emerging products for schizophrenia are expected to give tough market competition to Abilify, and the launch of late-stage emerging therapies will significantly impact the market[3].
Key Takeaways
- The global aripiprazole market is expected to grow at a CAGR of 5.1% from 2024 to 2030.
- The market is driven by increasing prevalence of mental health disorders, geriatric population, and rising healthcare expenditure.
- The loss of market exclusivity for Abilify Maintena in 2024 will lead to a decline in sales but will also increase accessibility due to generics.
- New developments like Abilify Asimtufii are expected to help Otsuka maintain its market position.
FAQs
Q: What is the current market size of the global aripiprazole market? A: The global aripiprazole market was valued at US$ 121 million in 2023[2].
Q: What is the projected CAGR for the global aripiprazole market from 2024 to 2030? A: The market is anticipated to grow at a CAGR of 5.1% during the forecast period[2].
Q: What is the impact of the loss of market exclusivity for Abilify Maintena? A: The loss of market exclusivity in October 2024 is expected to lead to a decline in sales due to the introduction of generic versions, with a projected decline from $1.5 billion in 2024 to $459 million by 2029[4].
Q: What new developments are Otsuka and Lundbeck introducing to the market? A: Otsuka and Lundbeck have received FDA approval for Abilify Asimtufii, a longer-acting version of the drug administered every two months[5].
Q: Who are the major players in the aripiprazole market? A: Key players include Otsuka Pharmaceutical, Teva, APOTEX, Barr Laboratories, Sun Pharmaceutical, and others[2].
Cited Sources
- Data Bridge Market Research - Global Aripiprazole Market - Industry Trends and Forecast to 2029
- Valuates Reports - Global Aripiprazole Drug Market Research Report 2024
- Research and Markets - ABILIFY MYCITE Drug Insight and Market Forecast - 2032
- Pharmaceutical Technology - Otsuka's Abilify Maintena: dual drug expiry signals sales decline
- Fierce Pharma - FDA signs off on Otsuka and Lundbeck's longer-acting Abilify
More… ↓


