Share This Page
Drug Sales Trends for VYTORIN
✉ Email this page to a colleague
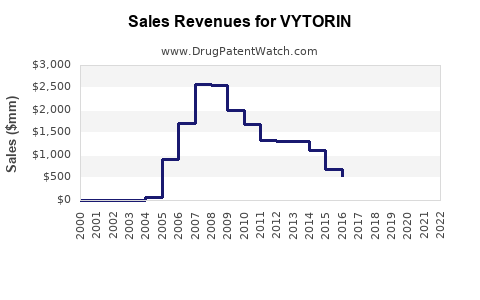
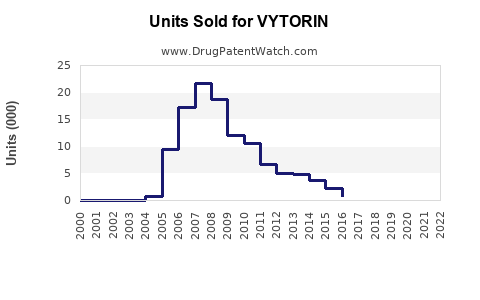
Annual Sales Revenues and Units Sold for VYTORIN
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| VYTORIN | ⤷ Try for Free | ⤷ Try for Free | 2021 |
| VYTORIN | ⤷ Try for Free | ⤷ Try for Free | 2020 |
| VYTORIN | ⤷ Try for Free | ⤷ Try for Free | 2019 |
| VYTORIN | ⤷ Try for Free | ⤷ Try for Free | 2018 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for Vytorin
Introduction to Vytorin
Vytorin is a combination medication that includes ezetimibe, a cholesterol absorption inhibitor, and simvastatin, a statin. This dual-action therapy is designed to reduce both the body's production of cholesterol and its absorption from the diet, making it a potent tool in managing hypercholesterolemia and reducing the risk of heart disease[4].
Market Drivers
Several key factors drive the demand for Vytorin in the global pharmaceutical market:
Rising Prevalence of Cardiovascular Diseases
The increasing global prevalence of cardiovascular diseases and hypercholesterolemia is a significant market driver. As lifestyle factors such as obesity, dietary habits, and stress contribute to higher cholesterol levels, the demand for effective cholesterol-lowering medications like Vytorin grows[4].
Trend Towards Combination Therapies
The trend towards combination therapies has gained traction in recent years. Vytorin's unique combination of ezetimibe and simvastatin offers a dual mechanism of action, which appeals to physicians seeking more potent treatment options for patients with stubbornly high cholesterol levels[4].
Personalized Medicine
There is a growing emphasis on personalized medicine, particularly as genetic testing becomes more accessible. Vytorin may find its niche as a tailored treatment for individuals with specific genetic cholesterol metabolism issues[4].
Regional Market Analysis
Global Market Overview
The global market for simvastatin, a key component of Vytorin, is expected to expand at a compound annual growth rate (CAGR) of 5.20% from 2023 to 2030. This growth is driven by the increasing demand for cholesterol management solutions worldwide[1].
North America
North America, particularly the United States, dominates the simvastatin market with a substantial 40% market share. The high prevalence of lifestyle-related diseases, robust healthcare infrastructure, and advanced diagnostic capabilities contribute to the widespread usage of simvastatin in this region. The adult segment accounts for the largest share of simvastatin usage in North America, driving sustained growth in the region[1].
Asia Pacific
The Asia Pacific region is the fastest-growing market for simvastatin, with a CAGR of 6.6% from 2024 to 2031. This growth is propelled by rising awareness about heart health, lifestyle changes leading to an increase in cardiovascular diseases, and improving healthcare infrastructure. The adult demographic is the primary driver of this growth, with increasing urbanization, dietary shifts, and stressful lifestyles elevating cholesterol-related concerns[1].
Sales Projections
Historical Sales
While specific historical sales data for Vytorin is not provided in the recent reports, it is known that Vytorin has been a significant product for Merck. For example, in 2010, Vytorin sales were projected at $1.96 billion, and by 2015, they had declined to $1.39 billion due to various market and competitive factors[3].
Future Projections
Given the growing demand for cholesterol management solutions and the trends towards combination therapies, Vytorin is expected to maintain a strong market presence. Here are some key projections:
-
Global Growth: The simvastatin market, which includes Vytorin, is expected to grow at a CAGR of 5.20% from 2023 to 2030. This indicates a steady increase in sales driven by global health trends and the need for effective cholesterol management[1].
-
Regional Performance: In North America, the market is expected to grow at a CAGR of 4.2%, while the Asia Pacific region will see a more rapid growth at a CAGR of 6.6%. These regional growth rates suggest that Vytorin will continue to be a significant player in these markets[1].
Manufacturing and Economic Insights
Project Economics
Setting up a manufacturing plant for ezetimibe/simvastatin (Vytorin) involves significant capital investments, detailed project funding, and thorough analysis of operating expenses. The project report by IMARC Group provides insights into unit operations, raw material requirements, utility requirements, and financial projections, including income and expenditure projections, taxation, depreciation, and liquidity analysis[4].
Profitability Analysis
The profitability of a Vytorin manufacturing plant is influenced by factors such as capital expenditure, operating expenditure, and expected ROI. The report also includes a detailed analysis of the regulatory procedures and approvals necessary for setting up such a plant[4].
Competitive Landscape
Vytorin competes in a market dominated by other cholesterol-lowering medications, including statins and combination therapies. The competitive landscape is influenced by factors such as patent expiries, generic competition, and the introduction of new therapies. For instance, the expiration of patents for leading statins has led to increased competition from generic alternatives, which can impact Vytorin's market share[5].
Key Takeaways
- Growing Demand: The global demand for cholesterol management solutions is increasing, driven by rising cardiovascular diseases and lifestyle changes.
- Regional Growth: North America and the Asia Pacific region are key markets for Vytorin, with the latter showing the fastest growth.
- Combination Therapy: Vytorin's dual mechanism of action makes it a preferred choice for managing hypercholesterolemia.
- Manufacturing Insights: Setting up a manufacturing plant for Vytorin involves detailed economic and regulatory analysis.
- Competitive Landscape: The market is competitive, with generic alternatives and new therapies impacting market dynamics.
FAQs
Q: What are the key components of Vytorin? A: Vytorin is a combination medication that includes ezetimibe, a cholesterol absorption inhibitor, and simvastatin, a statin.
Q: What drives the demand for Vytorin? A: The demand for Vytorin is driven by the rising prevalence of cardiovascular diseases, the trend towards combination therapies, and the need for personalized treatment options.
Q: Which regions are expected to see significant growth in Vytorin sales? A: North America and the Asia Pacific region are expected to see significant growth, with the Asia Pacific region being the fastest-growing market.
Q: What is the projected CAGR for the simvastatin market from 2023 to 2030? A: The simvastatin market is expected to grow at a CAGR of 5.20% from 2023 to 2030.
Q: What are the key factors to consider when setting up a manufacturing plant for Vytorin? A: Key factors include capital investments, project funding, operating expenses, regulatory procedures, and financial projections such as ROI and NPV.
Sources
- Cognitive Market Research: Global Simvastatin Drug Market Report 2024.
- Merck: Merck Announces Fourth-Quarter and Full-Year 2020 Financial Results.
- MM+M: Results may help Merck expand use of Vytorin.
- IMARC Group: Ezetimibe/Simvastatin (Vytorin) Manufacturing Plant Project Report 2024.
- Statista: Top pharma products by lifetime sales from launch to 2028.
More… ↓


