Introduction
XARELTO, known generically as rivaroxaban, is a prominent oral anticoagulant and Factor Xa inhibitor used in the treatment and prevention of various thrombotic conditions. Here, we delve into the market analysis and sales projections for XARELTO, highlighting its clinical significance, market dynamics, and future outlook.
Clinical Significance and Mechanism of Action
XARELTO is distinguished by its role as a selective inhibitor of Factor Xa, an enzyme crucial in the blood clotting process. By blocking Factor Xa, XARELTO reduces the levels of thrombin, thereby decreasing the risk of blood clots in veins and arteries. This mechanism is vital in treating conditions such as deep vein thrombosis, pulmonary embolism, and preventing strokes in patients with atrial fibrillation[4].
Market Size and Historical Growth
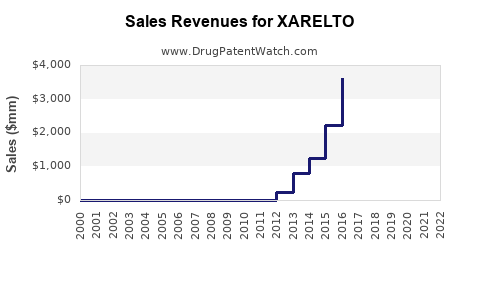
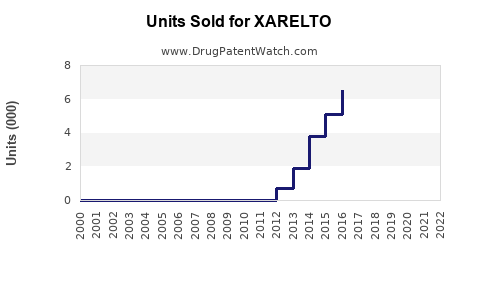
As of 2023, the global sales of XARELTO stood at USD 2,365 million. However, between 2020 and 2023, the sales growth was relatively flat, with a historical CAGR of 0.28%. This modest growth is attributed to various market and regulatory factors[4].
Current Market Dynamics
Sales Performance
XARELTO's sales have been impacted significantly by patent expirations. In the third quarter of 2024, sales of XARELTO declined by 23.0% on a currency- and portfolio-adjusted basis, primarily due to the loss of exclusivity and increased generic competition[2].
Geographic Market
North America is expected to dominate the rivaroxaban market due to the presence of major key players and a well-developed healthcare sector. However, the Asia-Pacific region is also anticipated to grow significantly during the forecast period, driven by increasing research and development activities and rising healthcare investments[3].
Forward-Looking Insights and Sales Forecasts
Projected Market Size
The global rivaroxaban market is projected to grow from USD 14.89 million in 2022 to USD 25.59 million by 2030, at a CAGR of 7.0% during the forecast period of 2023-2030. This growth is driven by factors such as the increasing number of hip and knee replacement surgeries, growing healthcare expenditure, and emerging markets[3].
Impact of Emerging Therapies
The market for XARELTO is expected to face competition from late-stage emerging therapies. However, XARELTO's established position and ongoing clinical developments, particularly in the treatment of Peripheral Artery Disease (PAD), are expected to support its market presence[1].
Regulatory and Clinical Developments
XARELTO's clinical and regulatory pathway continues to evolve. The drug has been a focal point for research in reducing the risk of major thrombotic vascular events in adult patients with PAD. Despite facing a boxed warning for potential bleeding risks from the FDA, XARELTO remains a critical therapy indicated for both coronary artery disease (CAD) and PAD post-lower extremity revascularization (LER)[1].
Challenges and Restraints
Patent Expirations
The loss of exclusivity for XARELTO is a significant challenge, leading to increased competition from generic versions. This is expected to continue impacting sales negatively into 2025 and beyond[2][5].
Side Effects and High Drug Costs
The high cost of XARELTO and its side effects, such as muscle pain, headache, dizziness, and others, are factors that will restrain market growth. These challenges necessitate careful management and mitigation strategies to maintain patient adherence and market share[3].
Strategic Positioning and Future Outlook
Bayer's Pharmaceutical Division
Bayer, one of the primary manufacturers of XARELTO, is confident in the division's ability to deliver at the upper end of the upgraded 2024 guidance. While XARELTO sales are declining, other products like Nubeqa™ and Kerendia™ are showing robust growth, which will help offset the impact of XARELTO's declining sales[2].
Market Expansion in PAD Treatment
The potential expansion of XARELTO in the treatment of PAD is a significant opportunity. The market analysis suggests that with advancements in treatment research and increasing healthcare allocations, XARELTO could maintain a strong market position in this segment[1].
Key Takeaways
- Market Growth: The global rivaroxaban market is expected to grow at a CAGR of 7.0% from 2023 to 2030.
- Sales Performance: XARELTO's sales are currently declining due to patent expirations but are expected to stabilize with new clinical developments.
- Geographic Focus: North America and the Asia-Pacific region are key markets for XARELTO.
- Challenges: High drug costs, side effects, and generic competition are significant challenges.
- Future Outlook: XARELTO's expansion in PAD treatment and ongoing clinical developments will be crucial for its market positioning.
FAQs
Q: What is the primary mechanism of action of XARELTO?
A: XARELTO works by inhibiting Factor Xa, an enzyme involved in the production of thrombin, thereby reducing the risk of blood clots.
Q: How has the sales performance of XARELTO been in recent years?
A: XARELTO's sales have declined recently due to patent expirations and increased generic competition, with a 23.0% decline in the third quarter of 2024.
Q: What are the key markets for XARELTO?
A: North America and the Asia-Pacific region are expected to be the dominant markets for XARELTO.
Q: What are the main challenges facing the XARELTO market?
A: High drug costs, side effects, and the impact of generic competition due to patent expirations are the main challenges.
Q: What is the projected market size for the global rivaroxaban market by 2030?
A: The global rivaroxaban market is projected to reach USD 25.59 million by 2030, growing at a CAGR of 7.0% from 2023 to 2030.
Sources
- ResearchAndMarkets.com, "Market Expansion for XARELTO in Peripheral Artery Disease Treatment through 2032: Forward-looking Insights and Sales Forecasts, Dynamics, and Emerging Therapies."
- Bayer, "Bayer: Strategic progress in challenging business environment."
- Data Bridge Market Research, "Global Rivaroxaban Market – Industry Trends and Forecast to 2030."
- GlobalData, "The Global Drug sales of Xarelto (2020 - 2026, USD Millions)."
- Fierce Pharma, "Bayer's 'transition year' looms as Xarelto sales falter."