Share This Page
Drug Sales Trends for ZETIA
✉ Email this page to a colleague
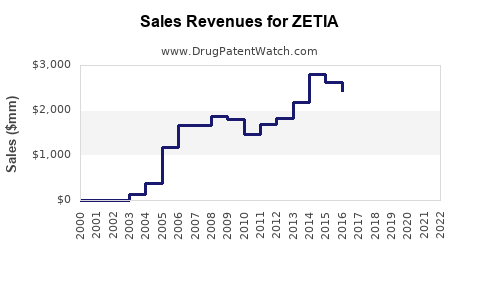
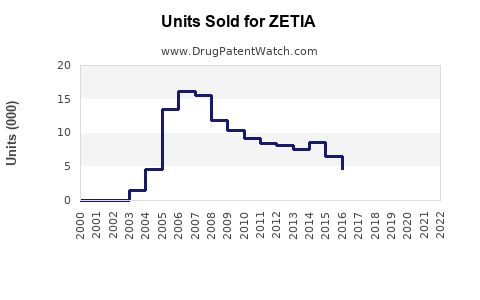
Annual Sales Revenues and Units Sold for ZETIA
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2021 |
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2020 |
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2019 |
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2018 |
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2017 |
| ZETIA | ⤷ Try for Free | ⤷ Try for Free | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Market Analysis and Sales Projections for ZETIA (Ezetimibe)
Introduction to ZETIA (Ezetimibe)
ZETIA, marketed under the generic name ezetimibe, is a lipid-lowering medication that has been a significant player in the management of hypercholesterolemia. Unlike traditional statins, ezetimibe works by reducing the absorption of dietary cholesterol in the small intestine, making it a valuable addition or alternative to statin therapy[3].
Current Market Position
As of 2016, ZETIA and its combination medication Vytorin were among Merck's top-selling drugs, expected to generate about $3.5 billion in sales. However, the loss of exclusivity for ZETIA marked a significant turning point. The first generic version of ZETIA was launched by Endo’s Par Pharmaceutical unit, followed by a 180-day exclusivity period before multiple generics could enter the market. This transition was anticipated to drastically reduce ZETIA's sales, with projections suggesting a drop to about $1.9 billion in 2017 and less than $1 billion by 2020[1].
Impact of Generic Competition
The introduction of generic versions of ZETIA has had a profound impact on Merck's revenue. The generics have not only eroded ZETIA's market share but also affected the sales of Vytorin, a combination medication that includes ezetimibe and a statin. The decline in sales is partly due to the higher margins associated with brand-name drugs compared to generics. Analysts have noted that much of ZETIA's growth since 2011 was driven by price hikes, which are no longer sustainable in a generic-dominated market[1].
Market Drivers and Trends
Despite the challenges posed by generic competition, the market for ezetimibe remains driven by several key factors:
- Increasing Prevalence of Cardiovascular Diseases: The rising incidence of cardiovascular diseases and high cholesterol levels due to obesity and unhealthy lifestyles continues to drive demand for effective cholesterol management medications[3].
- Combination Therapies: There is a growing trend towards combination therapies, with ezetimibe often used in conjunction with statins to achieve synergistic effects in lowering LDL cholesterol levels[3][4].
- Clinical Research and Expanded Indications: Ongoing research into the broader therapeutic applications of ezetimibe, such as preventing atherosclerosis progression, could expand its market further[3].
Sales Projections
The sales projections for ZETIA have been significantly impacted by the introduction of generics:
- 2017 Projections: Sales were expected to drop to about $1.9 billion, a substantial decline from the $3.5 billion in 2016[1].
- 2020 Projections: By 2020, sales were anticipated to fall below $1 billion, reflecting the continued erosion of market share by generic competitors[1].
Financial Impact on Merck
The loss of exclusivity for ZETIA has had a notable financial impact on Merck. While the company has seen growth in other areas, such as oncology and vaccines, the decline in ZETIA sales has been a significant factor in their financial performance. For instance, Merck's overall pharmaceutical sales have been driven by growth in oncology, vaccines, and hospital acute care, but this growth has been partially offset by declines in other areas, including the impact of generic competition on ZETIA[2].
Competitive Landscape
The cholesterol-fighting market has become increasingly crowded with the introduction of new therapies:
- PCSK9 Inhibitors: Drugs like Praluent and Repatha, although pricey, have entered the market. However, their high list prices have led payers to require patients to try ZETIA or Vytorin before approving the more expensive PCSK9 inhibitors. This has somewhat buffered the decline in ZETIA sales[1].
Key Takeaways
- Generic Competition: The introduction of generic versions of ZETIA has significantly reduced its sales and impacted Merck's revenue.
- Market Drivers: The market remains driven by the increasing prevalence of cardiovascular diseases, the trend towards combination therapies, and ongoing clinical research.
- Sales Projections: Sales are projected to continue declining as generic competition intensifies.
- Financial Impact: The decline in ZETIA sales has been offset partly by growth in other areas of Merck's portfolio.
FAQs
Q: What is the mechanism of action of ZETIA (ezetimibe)? A: ZETIA works by reducing the absorption of dietary cholesterol in the small intestine, unlike statins which inhibit cholesterol production in the liver[3].
Q: How has the introduction of generics affected ZETIA's sales? A: The introduction of generics has led to a significant decline in ZETIA's sales, with projections indicating a drop from $3.5 billion in 2016 to less than $1 billion by 2020[1].
Q: What are the key market drivers for ezetimibe? A: The key market drivers include the increasing prevalence of cardiovascular diseases, the trend towards combination therapies, and ongoing clinical research into broader therapeutic applications[3].
Q: How does ZETIA compare to PCSK9 inhibitors in terms of cost and usage? A: ZETIA is often required as a first-line treatment before payers approve the more expensive PCSK9 inhibitors like Praluent and Repatha[1].
Q: What is the impact of ZETIA's loss of exclusivity on Merck's financial performance? A: The loss of exclusivity has led to a decline in ZETIA sales, which has been partially offset by growth in other areas of Merck's portfolio, such as oncology and vaccines[2].
Sources
- FiercePharma: "Zetia generics launch sets Merck up for $1.4B hit to 2017 cholesterol sales"
- Merck: "Merck Announces Fourth-Quarter and Full-Year 2023 Financial Results"
- IMARC Group: "Ezetimibe (Zetia) Manufacturing Plant Report 2024"
- FDA: "ZETIA® - accessdata.fda.gov"
- Merck: "Merck Announces First-Quarter 2024 Financial Results"
More… ↓


