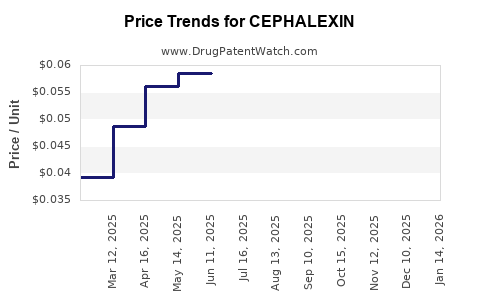
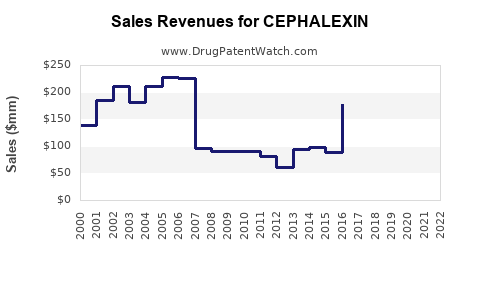
Cephalexin Market Dynamics and Financial Trajectory
Market Introduction
Cephalexin, a first-generation cephalosporin antibiotic, is widely used to treat various bacterial infections, including respiratory tract infections, skin and skin structure infections, bone infections, and genitourinary infections. It is also used to prevent endocarditis caused by bacterial infections[4].
Global Market Size and Growth
The global cephalexin API market is projected to expand significantly over the next few years. As of 2024, the market size is estimated to be USD XX million and is expected to grow at a compound annual growth rate (CAGR) of 6.30% from 2024 to 2031[1].
Regional Market Analysis
North America
North America holds a dominant position in the global cephalexin API market, accounting for around 40% of the global revenue. The region's stringent regulatory standards, emphasis on research and development, and strategic collaborations between pharmaceutical companies and research institutions drive market growth. The injection segment is particularly prominent due to its convenience and efficacy[1].
Europe
Europe accounts for approximately 30% of the global market size. The growing geriatric population, increased healthcare expenditure, and government initiatives to combat antimicrobial resistance are key drivers. The region's focus on healthcare quality and patient safety ensures consistent demand for high-quality APIs. The 99.5% purity segment holds the highest market share due to its ease of administration and regulatory compliance[1].
Latin America
Latin America contributes around 5% to the global revenue and is expected to grow at a CAGR of 5.7% from 2024 to 2031. The region's expanding healthcare infrastructure, rising awareness of infectious diseases, and favorable regulatory environment are significant factors. The 99.5% purity segment dominates due to its ease of administration and widespread availability[1].
Middle East and Africa
The Middle East and Africa region holds around 2% of the global market share and is projected to grow at a CAGR of 6.0% from 2024 to 2031. Increasing healthcare expenditure, government initiatives, and collaborations between pharmaceutical companies and healthcare organizations drive market expansion. The 99.5% purity segment is preferred for its established reputation for quality and efficacy[1].
Market Drivers
Increasing Prevalence of Bacterial Infections
The rising incidence of bacterial infections such as pneumonia, urinary tract infections, skin infections, and meningitis is a major driver. Cephalosporins, including cephalexin, are crucial in treating these infections due to their broad-spectrum activity and efficacy[3].
Advancements in Antibiotic Research
Continuous efforts in research and development to create more effective and resistant-free cephalosporins enhance the market's growth prospects. Innovations in drug formulations and delivery methods, such as advanced injection technologies, further support market expansion[3].
Healthcare Expenditure and Government Initiatives
Increasing healthcare expenditure and government initiatives to combat antimicrobial resistance and improve healthcare access are significant drivers. These initiatives often include funding for research, improving regulatory frameworks, and promoting the use of high-quality APIs[1][3].
Market Challenges
Cost Pressures and Pricing Flexibility
Cost pressures within healthcare systems pose a significant challenge. Pharmaceutical firms face pressure to lower drug prices and offer cost-effective treatments, limiting pricing flexibility and impacting profitability and research investments. Payer negotiations and reimbursement constraints exacerbate these challenges[1].
Availability and Accessibility
The availability of cephalexin in emerging nations is anticipated to hamper market growth. Limited access to healthcare facilities and economic constraints in these regions affect the demand for cephalexin[4].
Market Segmentation
By Formulation
The global cephalexin market is segmented into capsules, liquid suspension, and tablets. Each formulation has its own market share and growth prospects, with the injection segment being particularly prominent in regions like North America due to its convenience and efficacy[4].
By Distribution Channel
The market is segmented into retail pharmacies, online pharmacies, and hospital pharmacies. Hospital pharmacies often dominate due to the high demand for antibiotics in hospital settings[4].
Key Players and Strategies
Companies such as Lupin Pharmaceuticals Inc, PRAGMA, ALKEM LABS LTD, AUROBINDO PHARMA LTD, Hikma Pharmaceuticals PLC, Teva Pharmaceutical Industries Ltd, and Yung Shin Pharmaceutical Industrial Co are key players in the cephalexin market. These companies focus on strategy building models to strengthen their product portfolios and expand their business globally. Strategic collaborations and investments in R&D are common strategies to maintain market competitiveness[4].
Financial Trajectory
The financial trajectory of the cephalexin market is positive, driven by the growing demand for antibiotics and advancements in pharmaceutical technologies. Here are some key financial indicators:
- Global Market Size: Estimated at USD XX million in 2024, expected to grow at a CAGR of 6.30% from 2024 to 2031[1].
- Regional Growth: North America is expected to grow at a CAGR of 4.5%, Europe at 4.8%, Latin America at 5.7%, and the Middle East and Africa at 6.0% during the forecast period[1].
COVID-19 Impact
The COVID-19 pandemic has had a mixed impact on the cephalexin market. While it has accelerated the need for effective antibiotics due to secondary bacterial infections in COVID-19 patients, it has also disrupted supply chains and affected production capacities. However, the long-term growth prospects remain robust due to the ongoing need for antibiotics in treating various bacterial infections[5].
Key Takeaways
- The global cephalexin API market is expected to grow significantly, driven by increasing bacterial infections and advancements in antibiotic research.
- Regional markets such as North America, Europe, and Latin America are key contributors to the global market.
- Cost pressures and limited availability in emerging nations are significant challenges.
- Strategic collaborations and investments in R&D are crucial for market competitiveness.
- The market is segmented by formulation and distribution channel, with the injection segment and hospital pharmacies being prominent.
FAQs
Q: What is the expected CAGR of the global cephalexin API market from 2024 to 2031?
A: The global cephalexin API market is expected to grow at a CAGR of 6.30% from 2024 to 2031[1].
Q: Which region holds the largest market share in the global cephalexin API market?
A: North America holds around 40% of the global revenue and is the largest market for cephalexin API[1].
Q: What are the main drivers of the cephalexin market?
A: The main drivers include the increasing prevalence of bacterial infections, advancements in antibiotic research, and increasing healthcare expenditure and government initiatives[1][3].
Q: Which formulation segment dominates the cephalexin market?
A: The injection segment, particularly the 99.5% purity segment, dominates the market due to its convenience and efficacy[1][2].
Q: How has the COVID-19 pandemic impacted the cephalexin market?
A: The pandemic has accelerated the need for effective antibiotics but has also disrupted supply chains and affected production capacities. Long-term growth prospects remain robust[5].
Sources:
- Cognitive Market Research - Cephalexin API Market Report 2024 (Global Edition)
- Cognitive Market Research - Cefalexin API Market Report 2024 (Global Edition)
- Data Bridge Market Research - Global Cephalosporin Market – Industry Trends and Forecast to 2031
- The Insight Partners - Cephalexin Market Updates by 2031
- Stratview Research - Cephalosporin Market Size, Share, & Growth Analysis