Market Overview
Letrozole, an aromatase inhibitor, is a crucial drug in the treatment of hormone receptor-positive breast cancer in postmenopausal women. It works by lowering estrogen levels, which can slow or stop the growth of cancer cells. Here’s a detailed look at the market dynamics and financial trajectory of Letrozole.
Growth Rate and Market Size
The Letrozole market is expected to grow at a significant compound annual growth rate (CAGR) from 2024 to 2034. The market size is projected to reach USD billions by 2034, driven by an average growth pattern[1].
Drivers of Market Growth
Several factors are driving the growth of the Letrozole market:
Rising Incidence of Breast Cancer
The increasing incidence of breast cancer, particularly in postmenopausal women, is a major driver. This demographic shift contributes to a larger patient pool requiring Letrozole treatment[1].
Advancements in Oncology
Advancements in oncology and increased awareness of early cancer detection boost demand for Letrozole. Improved diagnostic techniques and treatment protocols enhance the drug's utility and acceptance[1].
Off-Label Use
Letrozole's off-label use in fertility treatments, especially for women with polycystic ovary syndrome (PCOS), adds to its market growth. This dual application expands its user base beyond cancer treatment[1].
Increased Healthcare Spending and Government Initiatives
Increased healthcare spending and government initiatives aimed at improving cancer treatment and accessibility further drive the market. The availability of generic versions due to patent expirations makes Letrozole more accessible and affordable[1].
Regional Analysis
North America
North America dominates the Letrozole market, accounting for more than 46% of the market share. The high prevalence of hormone receptor-positive breast cancer, advanced healthcare systems, and significant investment in cancer research and treatment in this region contribute to its leading position[1].
Europe
Europe's Letrozole market benefits from a robust healthcare infrastructure and a high incidence of breast cancer, particularly in Western Europe. Countries like Germany, France, and the UK have extensive cancer treatment facilities and strong healthcare systems. However, Eastern Europe faces challenges related to healthcare access and economic disparities[1].
Asia Pacific
The Asia-Pacific region is experiencing rapid growth due to rising cancer rates and improving healthcare infrastructure. Countries like China and India are major contributors, driven by increasing awareness, enhanced healthcare facilities, and government initiatives aimed at improving cancer treatment and accessibility[1].
Key Market Players
The Letrozole market is dominated by several large pharmaceutical companies, including:
- Novartis AG
- Sun Pharmaceutical Industries Ltd.
- Teva Pharmaceuticals
- Intas
- Cipla
- Medgrow Health Care
- Natco Pharma Ltd
- Aristo Pharmaceutical Pvt Ltd[1].
Financial Performance
Revenue and Market Share
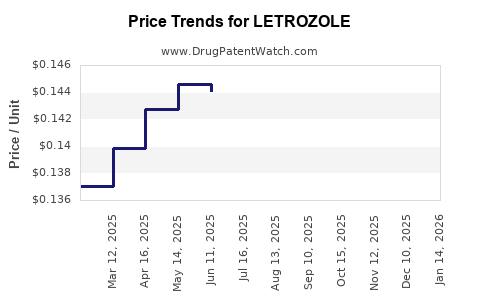
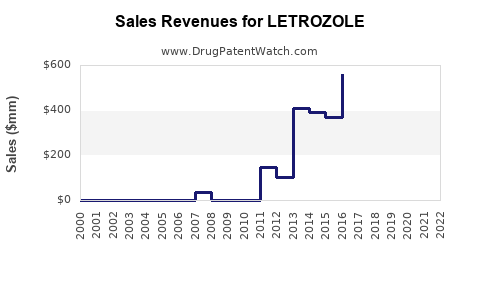
The market revenue for Letrozole is expected to increase significantly over the forecast period. The drug's widespread use in both branded and generic forms contributes to its revenue growth. Hospital pharmacies, retail pharmacies, online pharmacies, and specialty clinics are key distribution channels[1].
Cost-Effectiveness
Studies have shown that Letrozole is cost-effective compared to other hormonal therapies. For instance, a cost-effectiveness analysis found that first-line hormonal therapy with Letrozole gains additional life years at a cost of £5,917, and the cost per additional quality-adjusted life year (QALY) gained is £8,514[3].
Combination Therapies
The use of Letrozole in combination with other drugs, such as Pfizer's CDK 4/6 inhibitor Palbociclib, has shown significant benefits in prolonging progression-free survival in patients with advanced breast cancer. This combination therapy further enhances the financial trajectory of Letrozole by expanding its therapeutic applications[5].
Market Segments
Type and Application
The Letrozole market is segmented based on type (tablet, capsule, solution) and application (breast cancer treatment, fertility treatment). These segments are crucial for understanding the diverse uses and market dynamics of Letrozole[4].
Geographical Segments
The market is also segmented geographically into North America, Europe, Asia-Pacific, South America, and the Middle East and Africa. Each region has its unique market dynamics influenced by local healthcare systems, regulatory environments, and patient demographics[4].
Challenges and Opportunities
Generic Competition
The availability of generic versions of Letrozole due to patent expirations presents both opportunities and challenges. While generics make the drug more accessible, they also increase competition and can impact the revenue of branded versions[1].
Regulatory Environment
Government initiatives and regulatory changes can significantly impact the market. For example, favorable regulatory environments can facilitate the approval and distribution of Letrozole, while stringent regulations can pose challenges[1].
Research and Development
Continuous research and development in oncology and fertility treatments offer opportunities for Letrozole to be used in new therapeutic areas or in combination with other drugs, further expanding its market potential[5].
Key Takeaways
- The Letrozole market is expected to grow significantly due to the rising incidence of breast cancer and advancements in oncology.
- North America dominates the market, followed by Europe and the Asia-Pacific region.
- Key market players include Novartis AG, Sun Pharmaceutical Industries Ltd., and Teva Pharmaceuticals.
- Letrozole is cost-effective and gains additional life years and QALYs compared to other hormonal therapies.
- The drug's use in combination therapies and off-label applications in fertility treatments further enhance its market growth.
FAQs
What is Letrozole used for?
Letrozole is primarily used to treat hormone receptor-positive breast cancer in postmenopausal women by lowering estrogen levels. It is also used off-label to induce ovulation in women with fertility issues.
Which region dominates the Letrozole market?
North America dominates the Letrozole market with a share of more than 46%.
What are the key drivers of the Letrozole market?
Key drivers include the rising incidence of breast cancer, advancements in oncology, off-label use in fertility treatments, and increased healthcare spending and government initiatives.
Who are the major players in the Letrozole market?
Major players include Novartis AG, Sun Pharmaceutical Industries Ltd., Teva Pharmaceuticals, Intas, Cipla, Medgrow Health Care, Natco Pharma Ltd, and Aristo Pharmaceutical Pvt Ltd.
Is Letrozole cost-effective?
Yes, Letrozole is cost-effective. It gains additional life years and QALYs at a reasonable cost compared to other hormonal therapies.
Sources
- We Market Research: Letrozole Market Size, Share, Trends Analysis & Forecast.
- Laboratorios Farmaceuticos ROVI: Results_Press Release.
- PubMed: A trial-based cost-effectiveness analysis of letrozole followed by tamoxifen versus tamoxifen followed by letrozole for postmenopausal women with advanced breast cancer.
- Market Research Intellect: Global Letrozole API Sales Market Size, Scope And Forecast Report.
- Pfizer: Pfizer’s Novel CDK 4/6 Inhibitor Palbociclib plus Letrozole Significantly Prolonged Progression-Free Survival in Patients with Advanced Breast Cancer.